题目内容
有一薄壁玻璃瓶,在温度恒定情况下,称量质量三次:(1)瓶抽空了;(2)瓶内装有一个大气压的空气;(3)瓶内装满1.5个大气压的未知气体.三次称量的数值是: .试计算未知气体的相对分子质量.已知空气的相对分子质量为29.瓶壁的体积与瓶内的容积相比可以忽略不计.
.试计算未知气体的相对分子质量.已知空气的相对分子质量为29.瓶壁的体积与瓶内的容积相比可以忽略不计.
 .试计算未知气体的相对分子质量.已知空气的相对分子质量为29.瓶壁的体积与瓶内的容积相比可以忽略不计.
.试计算未知气体的相对分子质量.已知空气的相对分子质量为29.瓶壁的体积与瓶内的容积相比可以忽略不计.43.5
设容器的体积为V,并设将未知气体转化成一个大气压时的体积为
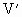 ,大气压强为
,大气压强为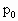 ,依题意可知:装入的空气质量为
,依题意可知:装入的空气质量为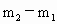 ,装入的未知气体的质量为
,装入的未知气体的质量为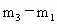 .
.对于未知气体有:1.5
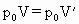
则未知气体在一个大气压下且体积为V时所具有的质量为:
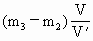
所以该气体的相对分子量为:
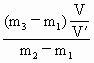 ×29=43.5
×29=43.5
练习册系列答案
相关题目

 光滑斜面上,气缸A倒扣在水平地面上,气缸A和B内装有相同质量的同种气体,体积都为
光滑斜面上,气缸A倒扣在水平地面上,气缸A和B内装有相同质量的同种气体,体积都为 ,温度都为
,温度都为 ,如图所示,此时气缸A的气缸筒恰好对地面没有压力.设气缸内气体的质量远小于活塞的质量,大气对活塞的压力等于活塞重的1.5倍.
,如图所示,此时气缸A的气缸筒恰好对地面没有压力.设气缸内气体的质量远小于活塞的质量,大气对活塞的压力等于活塞重的1.5倍.
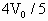 ,气缸B内的气体的温度是多少?
,气缸B内的气体的温度是多少?
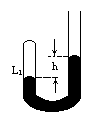
 时,左、右管内液面高度相等,两管内空气柱长度均为l,压强均为大气压强
时,左、右管内液面高度相等,两管内空气柱长度均为l,压强均为大气压强 .现使两边温度同时逐渐升高,求:
.现使两边温度同时逐渐升高,求: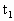 =39℃时,管内水银封闭着
=39℃时,管内水银封闭着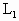 =8cm长的空气柱,管内两水银面高度差h=2cm.现在给气柱加热,使气柱长变为
=8cm长的空气柱,管内两水银面高度差h=2cm.现在给气柱加热,使气柱长变为 =9cm,求这时气体的温度
=9cm,求这时气体的温度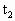 .
.

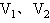 的两容器,用一根不导热的细管(体积可忽略不计)连通,如图所示.容器内充满空气,最初两容器都浸在温度为
的两容器,用一根不导热的细管(体积可忽略不计)连通,如图所示.容器内充满空气,最初两容器都浸在温度为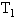 的水中,然后把容积为
的水中,然后把容积为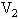 的容器用温度为
的容器用温度为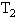 的水蒸汽包围,但容积为
的水蒸汽包围,但容积为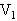 的容器仍保持原来温度.如果两个容器内空气的最初压强都为
的容器仍保持原来温度.如果两个容器内空气的最初压强都为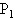 ,求最后两容器内空气的压强
,求最后两容器内空气的压强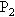 .(设容器的膨胀忽略不计)
.(设容器的膨胀忽略不计)