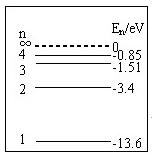
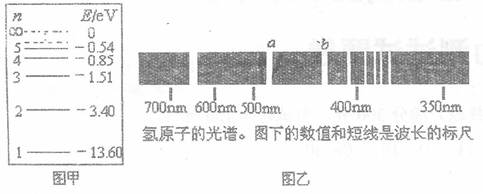
题目内容
(1)有一群氢原子处在量子数n=3的激发态,画一能级图,在图上用箭头标明这些氢原子能发出几种光谱线.
(2)计算这几条光谱线中最短的波长.(已知En =E1/n2,普朗克常量为6.63×10-34J.s )
(1)
(2) 165 nm.
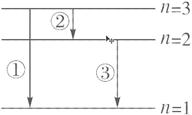
(1)氢原子一共发出3种光谱线,如右图所示:

(2)从n=3的激发态跃迁到基态时,放出的光子能量最大,波长最短.
由E3-E1=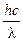 得:
得:
λ=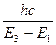 =
=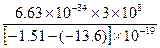 m
m
=1.65×10-7 m="165" nm.

(2)从n=3的激发态跃迁到基态时,放出的光子能量最大,波长最短.
由E3-E1=
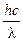 得:
得:λ=
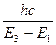 =
=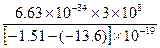 m
m=1.65×10-7 m="165" nm.

练习册系列答案
 作业辅导系列答案
作业辅导系列答案 同步学典一课多练系列答案
同步学典一课多练系列答案
相关题目
 、
、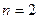 的轨道上运动时,下列相关物理量的关系正确的有 ( )
的轨道上运动时,下列相关物理量的关系正确的有 ( )
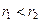
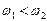
 ,现有大量的氢原子处于n=4的激发态,当向低能级跃迁时辐射出若干不同频率的光。关于这些光下列说法正确的是( )
,现有大量的氢原子处于n=4的激发态,当向低能级跃迁时辐射出若干不同频率的光。关于这些光下列说法正确的是( )