题目内容
已知铜的摩尔质量M=63.5g/mol,铜的密度是ρ=8.9g/cm3,试估算铜原子的质量和铜原子的体积.已知NA=6.02×1023mol-1.
1.06×10-22g,1.2×10-23cm3
设想铜原子是一个个紧挨着排列的,利用阿伏加德罗常数即得.
铜原子的质量:
m=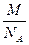 =
=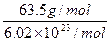 =1.06×10-22g.
=1.06×10-22g.
铜原子的体积:
v=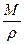 ·
·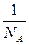 =
=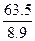 ×
×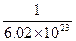 cm3=1.2×10-23cm3.
cm3=1.2×10-23cm3.
铜原子的质量:
m=
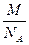 =
=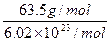 =1.06×10-22g.
=1.06×10-22g.铜原子的体积:
v=
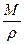 ·
·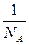 =
=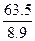 ×
×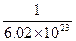 cm3=1.2×10-23cm3.
cm3=1.2×10-23cm3.
练习册系列答案
相关题目
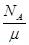
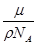

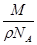
 ,摩尔质量M=
,摩尔质量M= .试估算金刚石内碳原子间的平均距离(结果保留两位有效数字).
.试估算金刚石内碳原子间的平均距离(结果保留两位有效数字).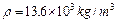

 的某种油,用滴管滴出一滴油在水面上形成油膜,已知这滴油的体积为V=0.5×10-3cm3,形成的油膜面积为S=0.7m2,油的摩尔质量M=0.9kg/mol,若把油膜看成是单分子层,每个油分子看成球形,那么:(1)油分子的直径是多少?(2)由以上数据可粗略测出阿伏加德罗常数NA是多少?(先列出计算式,再代入数据计算,只要求保留一位有效数字)
的某种油,用滴管滴出一滴油在水面上形成油膜,已知这滴油的体积为V=0.5×10-3cm3,形成的油膜面积为S=0.7m2,油的摩尔质量M=0.9kg/mol,若把油膜看成是单分子层,每个油分子看成球形,那么:(1)油分子的直径是多少?(2)由以上数据可粗略测出阿伏加德罗常数NA是多少?(先列出计算式,再代入数据计算,只要求保留一位有效数字)