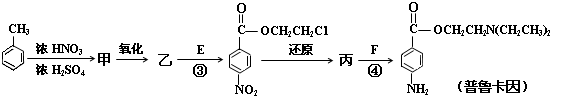
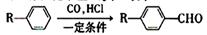
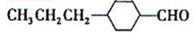��Ŀ����
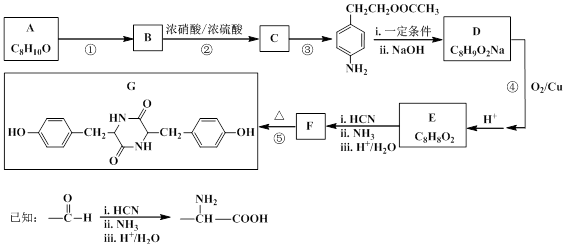
A��B��C��D���ַ����廯���ﶼ��ijЩֲ��ӷ����е���Ҫ�ɷ֣��е���ҩ��е������ϡ����ǵĽṹ��ʽ������ʾ��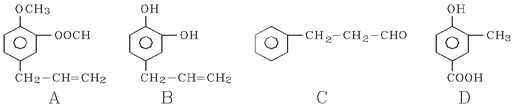
��ش��������⣺
��1��1 mol B���뺬 molBr2����ˮ��Ӧ��C�й����ŵ����� ��
��2���ܷ���������Ӧ����___________����A��B��C��D��գ���
��3����D��һ��ͬ���칹��G�Ľṹ��ʽ���£�д��G������NaOH��Һ���ȷ�Ӧ�Ļ�ѧ���̣� ��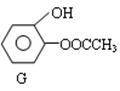
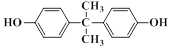
��D����һ��ͬ���칹��H���䱽�������������ڵ�ȡ������������ʹFeCl3��Һ����ɫ��������NaHCO3��Һ��Ӧ�ų�CO2���壬H�Ľṹ��ʽ�� ��
��4������ͼ��C��һ����Ӧ������E��E��B��ͬ���칹�壬��Ӧ������_______________ ���Ӧ�������ƣ���д��F�Ľṹ��ʽ_______________________��
��1��4 ��2�֣� ȩ�� ��2�֣�
��2��A��C ��2�֣� ��3����  ��3�֣�
��3�֣�
�� ��2�֣�
��2�֣�
��4��������Ӧ ��2�֣�  ��3�֣�
��3�֣�
���������������1������B�Ľṹ��֪�����ǻ����ڶ�λ�����巢��ȡ����̼̼˫�����巢���ӳɷ�Ӧ�����������4molBr2,C�еĹ�����Ϊȩ�����Ʒ����к���ȩ���IJ��ܷ���������Ӧ���ʴ�ΪA��C���Ǣ� ��
�� ��C��һ����Ӧ������E��E��B��ͬ���칹�壬��Ӧ��Ϊ������Ӧ��EΪ����Ӧ��Ϊ������Ӧ��F�Ľṹ��ʽΪC6H5CH2CH2COOCH2CH3.
��C��һ����Ӧ������E��E��B��ͬ���칹�壬��Ӧ��Ϊ������Ӧ��EΪ����Ӧ��Ϊ������Ӧ��F�Ľṹ��ʽΪC6H5CH2CH2COOCH2CH3.
���㣺�����л���Ľṹ�����ʵ����֪ʶ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���������Ǿ��п������Ϳ��������óɷ֣��������Ҳ��ͨ��ͼ��ʾ�����ϳɣ�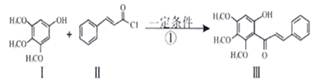
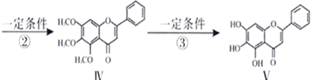
��֪ ��
��
��1����Ӧ�ٵ�����
��2�������囹�ķ���ʽ
��3����A�Ľṹ��ʽ ��
��1molV�������NaOH ��1mol������������H2 mol��1�֣�
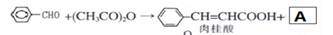
��4��������I�͢�Ӧ�����Եõ�һ�������÷�Ӧ�Ļ�ѧ����ʽ�� ��
��5�����ڢ���˵����ȷ����( )
| A������������������ |
| B���ܷ���ˮ�ⷴӦ |
| C��ʹ����KMnO4��Һ��ɫ |
| D����FeCl3������ɫ��Ӧ |
�ٷ����к��б��� ���Ȼ��뱽��ֱ������
 ���������
��������� ���ۺϵõ�����д���þ�̼�����Ľṹ��ʽ______________________________��
���ۺϵõ�����д���þ�̼�����Ľṹ��ʽ______________________________��
 ��
��