��Ŀ����
 �ס�������ͬѧ�ֱ��ͭ����������ķ�Ӧ����̽����������벢��ɶ��й�����Ľ��
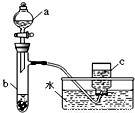
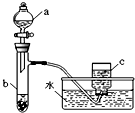
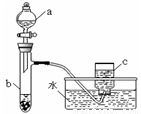
�ס�������ͬѧ�ֱ��ͭ����������ķ�Ӧ����̽����������벢��ɶ��й�����Ľ����1������ͬѧ��������ͼ��ʾװ����֤ͭ������ķ�Ӧ����֪����a��ʢ������һ��Ũ�ȵ�������Һ���Թ�b��ʢ��3.2gͭ��
����д��ͭ��ϡ���ᷴӦ�����ӷ�Ӧ����ʽ
3Cu+8H++2NO3-�T3Cu2++4H2O+2NO��
3Cu+8H++2NO3-�T3Cu2++4H2O+2NO��
������ͭ������ķ�Ӧ�У����������ֳ��Ļ�ѧ������Ҫ��
���Ժ�ǿ������
���Ժ�ǿ������
��������c���ռ���������Ҫ��
NO
NO
���������ʽ���ܴ��Թ�b�з�Ӧ������ȫ�������b��Һ���������������������Һ�������ɳ�����������
4.9
4.9
g����2������ͬѧ������ϡ����ķ�Ӧ����̽����������a mol Fe�ͺ���b mol HNO3��ϡ�������ʵ�飬����������ǡ�÷�Ӧ����ʣ�࣬��HNO3ֻ����ԭ��NO����Ӧ��������Һ�к���Fe3+��Fe2+����
�ٷ�Ӧ���������Һ��NO3-�����ʵ�����ȡֵ��Χ��
2amol��n��NO3-����3amol
2amol��n��NO3-����3amol
�����ú�a�Ĵ���ʽ�ȱ�ʾ��������Ӧ���������Һ��Fe3+��Fe2+�����ʵ���֮��n��Fe3+����n��Fe2+�� Ϊ3��1����b��a��ֵΪ
| 11 |
| 3 |
| 11 |
| 3 |
��������1����ͭ��ϡ���ᷴӦ��������ͭ��һ��������ˮ���ݴ���д��
�ڷ�Ӧ��NԪ�صĻ��ϼ۲���δ�仯��������ͭ�����ֽ������ɵ��������
�۽�������c������ΪNO2��NO2��ˮ��Ӧ����NO��
��ͭ��ȫ��Ӧ��������ͭ����b��Һ���������������������Һ��ͭ������ȫת��Ϊ������ͭ����������n=
����ͭ�����ʵ���������ͭԭ���غ����������ͭ�����ʵ������ٸ���m=nM���㣻
��2�������ü����������ȫ��ΪFe3+ʱ����Һ��NO3-�����ʵ������ȫ��ΪFe2+ʱ����Һ��NO3-�����ʵ�����С���ݴ˽��
����a��ʾ����Һ��Fe3+��Fe2+�����ʵ�����������a��ʾ����Һ��NO3-�����ʵ��������ݵ�ԭ���غ���a��b��ʾ��NO�����ʵ��������ݵ���ת���غ���
�ڷ�Ӧ��NԪ�صĻ��ϼ۲���δ�仯��������ͭ�����ֽ������ɵ��������
�۽�������c������ΪNO2��NO2��ˮ��Ӧ����NO��
��ͭ��ȫ��Ӧ��������ͭ����b��Һ���������������������Һ��ͭ������ȫת��Ϊ������ͭ����������n=
| m |
| M |
��2�������ü����������ȫ��ΪFe3+ʱ����Һ��NO3-�����ʵ������ȫ��ΪFe2+ʱ����Һ��NO3-�����ʵ�����С���ݴ˽��
����a��ʾ����Һ��Fe3+��Fe2+�����ʵ�����������a��ʾ����Һ��NO3-�����ʵ��������ݵ�ԭ���غ���a��b��ʾ��NO�����ʵ��������ݵ���ת���غ���
����⣺��1����ͭ��ϡ���ᷴӦ��������ͭ��һ��������ˮ����Ӧ���ӷ���ʽΪ��3Cu+8H++2NO3-�T3Cu2++4H2O+2NO����
�ʴ�Ϊ��3Cu+8H++2NO3-�T3Cu2++4H2O+2NO����
�ڷ�Ӧ��NԪ�صĻ��ϼ۲���δ�仯��������ͭ����������ᣬ�Բ��ֽ������ɵ���������������ǿ�����ԣ�
�ʴ�Ϊ�����Ժ�ǿ�����ԣ�
�۽�������c������ΪNO2��NO2��ˮ��Ӧ����NO������c���ռ���������Ҫ��NO���ʴ�Ϊ��NO��
��3.2gͭ�����ʵ���Ϊ
=0.05mol����b��Һ���������������������Һ��ͭ������ȫת��Ϊ������ͭ����������ͭԭ���غ��֪n[Cu��OH��2]=n��Cu��=0.05mol����m[Cu��OH��2]=0.05mol��98g/mol=4.9g���ʴ�Ϊ��4.9��
��2����ȫ��ΪFe3+ʱ����Һ��NO3-�����ʵ������NO3-�����ʵ������Ϊ3amol��ȫ��ΪFe2+ʱ����Һ��NO3-�����ʵ�����С����NO3-�����ʵ�����СΪ3amol��������Һ�к���Fe3+��Fe2+����2amol��n��NO3-����3amol��
�ʴ�Ϊ��2amol��n��NO3-����3amol��
��n��Fe3+����n��Fe2+��=3��1����n��Fe3+��=
amol��n��Fe2+��=
amol������Һ��n��NO3-��=3n��Fe3+��+2n��Fe2+��=
amol��3+
amol��2=
mol���ɵ�ԭ���غ��֪��n��NO��=bmol-
mol�����ݵ���ת���غ��У�
amol��3+
amol��2=��b-
��mol����5-2����������b��a=
��
�ʴ�Ϊ��
��
�ʴ�Ϊ��3Cu+8H++2NO3-�T3Cu2++4H2O+2NO����
�ڷ�Ӧ��NԪ�صĻ��ϼ۲���δ�仯��������ͭ����������ᣬ�Բ��ֽ������ɵ���������������ǿ�����ԣ�
�ʴ�Ϊ�����Ժ�ǿ�����ԣ�
�۽�������c������ΪNO2��NO2��ˮ��Ӧ����NO������c���ռ���������Ҫ��NO���ʴ�Ϊ��NO��
��3.2gͭ�����ʵ���Ϊ
| 3.2g |
| 64g/mol |
��2����ȫ��ΪFe3+ʱ����Һ��NO3-�����ʵ������NO3-�����ʵ������Ϊ3amol��ȫ��ΪFe2+ʱ����Һ��NO3-�����ʵ�����С����NO3-�����ʵ�����СΪ3amol��������Һ�к���Fe3+��Fe2+����2amol��n��NO3-����3amol��
�ʴ�Ϊ��2amol��n��NO3-����3amol��
��n��Fe3+����n��Fe2+��=3��1����n��Fe3+��=
| 3 |
| 4 |
| 1 |
| 4 |
| 3 |
| 4 |
| 1 |
| 4 |
| 11a |
| 4 |
| 11a |
| 4 |
| 3 |
| 4 |
| 1 |
| 4 |
| 11a |
| 4 |
| 11 |
| 3 |
�ʴ�Ϊ��
| 11 |
| 3 |
���������⿼������Ļ�ѧ���ʡ���ѧ����ȣ��Ѷ��еȣ�ע�������غ�˼���뼫���Ľ��еļ��㣮

��ϰ��ϵ�д�
�����Ŀ
 2CaO+2S02��+O2����
2CaO+2S02��+O2����
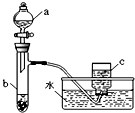 �ס�������ͬѧ�ֱ��ͭ����������ķ�Ӧ����̽����������벢��ɶ��й�����Ľ��
�ס�������ͬѧ�ֱ��ͭ����������ķ�Ӧ����̽����������벢��ɶ��й�����Ľ��