��Ŀ����
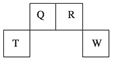
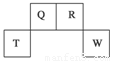
(8��)������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ����ͼ��ʾ�����У�T��������������������������ȡ���ش��������⣺
(1)T��ԭ�ӽṹʾ��ͼΪ________��
(2)Ԫ�صķǽ�����(ԭ�ӵĵõ�������)��Q________W(�ǿ�ڡ������ڡ�)��
(3)W�ĵ�����������������ˮ����Ũ��Һ�����ܷ�����Ӧ�������������ʣ�����һ�������壬��Ӧ�Ļ�ѧ����ʽΪ__________________________��
(4)ԭ��������R��1��Ԫ�ص�һ���⻯���ֽܷ�Ϊ������һ���⻯��˷ֽⷴӦ�Ļ�ѧ����ʽ��__________________��
(5)R�ж�����������м���Է���������С����һ�������£�2 L�ļ�������0.5 L���������ϣ����û�����屻������NaOH��Һ��ȫ���պ�û�����������������R�ĺ�������ֻ��һ�֣���ú������εĻ�ѧʽ��________________��
(6)��298K�£�Q��T�ĵ��ʸ�1 mol��ȫȼ�գ��ֱ�ų�����a kJ��b kJ����֪һ�������£�T�ĵ����ܽ�Q������������������û������������û���Ӧ����3 mol Q�ĵ��ʣ���÷�Ӧ��298K�µĦ�H��________(ע���������浥�ʾ�Ϊ���ȶ�����)��
(1)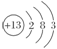 ��
��
(2)����
(3)S��2H2SO4(Ũ)3SO2����2H2O
(4)2H2O22H2O��O2��(������������)
(5)NaNO2(6)(3a��4b) kJ/mol
��������