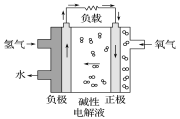
ĚâÄżÄÚČÝ
ˇľĚâÄżˇżÄłĘµŃéС×éĹäÖĆ0.10mol/LNaOHČÜŇş˛˘˝řĐĐÓĐąŘĐÔÖĘʵŃ飬»Ř´đĎÂÁĐÎĘĚ⡣
Ł¨1Ł©ČôʵŃéÖĐ´óÔĽŇŞĘąÓĂ475mLNaOHČÜŇşŁ¬ÖÁÉŮĐčŇŞłĆÁżNaOHąĚĚĺ______gˇŁ
Ł¨2Ł©´ÓĎÂÍĽÖĐѡÔńłĆÁżNaOHąĚĚĺËůĐčŇŞµÄŇÇĆ÷ĘÇŁ¨Ěî×ÖĸŁ©____________ˇŁÔÚĹäÖĆČÜҺʱŁ¬µ±żěµ˝´ďżĚ¶ČĎßʱČçşÎ˝řĐж¨ČݲŮ×÷_____________________ˇŁ
ĂűłĆ | ÍĐĹĚĚěĆ˝ Ł¨´říŔÂ룩 | СÉŐ± | ŰáŰöÇŻ | ˛ŁÁ§°ô | Ň©ł× | ÁżÍ˛ |
ŇÇĆ÷ |
|
|
|
|
|
|
ĐňşĹ | a | b | c | d | e | f |
Ł¨3Ł©ĎÂÁĐÇéżö»áĘąËůĹäČÜҺŨ¶ČĆ«µÍµÄĘÇŁ¨ĚîĐňşĹŁ© ________________
˘ŮłĆÁż¶ÁĘýʱŁ¬×óĹ̸ߣ¬ÓŇĹ̵Í
˘ÚËůÓõÄíŔÂëÉúĐâ
˘ŰČÜҺתŇƵ˝ČÝÁżĆżşóŁ¬Î´˝řĐĐĎ´µÓ˛Ů×÷
˘ÜתŇĆČÜҺǰČÝÁżĆżÄÚÓĐÉŮÁżŐôÁóË®
˘Ý¶¨ČÝʱŁ¬ŃöĘÓČÝÁżĆżµÄżĚ¶ČĎß
˘ŢÔÚÉŐ±ÖĐČÜ˝âNaOHşóŁ¬Á˘Ľ´˝«ËůµĂČÜҺעČëČÝÁżĆżÖĐ
˘ß¶¨ČÝşóҡÔČŁ¬·˘ĎÖŇşĂ潵µÍŁ¬ÓÖ˛ąĽÓÉŮÁżË®Ł¬ÖŘĐ´ﵽżĚ¶ČʵŃéĘŇÓűĹäÖĆ
˘ŕÓĂÁżÍ˛ÁżČˇŇ»¶¨ÁżĹ¨ÁňËáĹäÖĆϡÁňËᣬתŇƵ˝ÉŐ±şóŁ¬ÓĂÉŮÁżË®Ď´µÓÁżÍ˛
ˇľ´đ°¸ˇż2.0abeµ±ŇşĂćľÝżĚ¶ČĎß1-2cmʱ¸ÄÓĂ˝şÍ·µÎąÜĽÓË®ÖÁČÜŇş°ĽĂćÓëżĚ¶ČĎßĎŕÇТ٢ۢݢß
ˇľ˝âÎöˇż
(1)¸ůľÝĹäÖĆČÜŇşĚĺ»ýѡÔńşĎĘĘČÝÁżĆż,¸ůľÝm=cVˇÁMĽĆËăĐčŇŞÇâŃő»ŻÄƵÄÖĘÁżŁ»
(2)łĆÁżŇ»¶¨ÖĘÁżµÄąĚĚĺÎďÖĘŇ»°ăÓĂÍĐĹĚĚěĆ˝ˇ˘Ň©ł×Ł¬łĆÁż¸ŻĘ´ĐÔŇ©Ć·Ó¦ÓĂСÉձʢ·ĹŁ»
(3)·ÖÎö˛Ů×÷¶ÔČÜÖʵÄÎďÖʵÄÁżşÍČÜŇşĚĺ»ýµÄÓ°Ďě,¸ůľÝc=n/V˝řĐĐÎó˛î·ÖÎöŁ»
(1)ĹäÖĆ475mLˇ˘0.10mol/LNaOHČÜŇş,ӦѡÔń500mL ČÝÁżĆż,ʵĽĘĹäÖĆ500mLČÜŇş,ĐčŇŞÇâŃő»ŻÄĆÖĘÁżm=0.5ˇÁ0.1ˇÁ40=2.0gŁ»Ňň´ËŁ¬±ľĚâŐýČ·´đ°¸ĘÇŁş2.0ˇŁ
(2)ÇâŃő»ŻÄĆľßÓиŻĘ´ĐÔËůŇÔłĆÁżÇâŃő»ŻÄĆąĚĚĺÓ¦ÓõÄŇÇĆ÷:ÍĐĹĚĚěĆ˝,Ň©ł×,СÉŐ±Ł»ÔÚĹäÖĆČÜҺʱŁ¬µ±ŇşĂćľÝżĚ¶ČĎß1-2cmʱ¸ÄÓĂ˝şÍ·µÎąÜĽÓË®ÖÁČÜŇş°ĽĂćÓëżĚ¶ČĎßĎŕÇĐŁ»Ňň´ËŁ¬ŐýČ·´đ°¸ŁşabeŁ»µ±ŇşĂćľÝżĚ¶ČĎß1-2cmʱ¸ÄÓĂ˝şÍ·µÎąÜĽÓË®ÖÁČÜŇş°ĽĂćÓëżĚ¶ČĎßĎŕÇСŁ
(3)˘ŮłĆÁż¶ÁĘýʱ,×óĹ̸ß,ÓŇĹ̵Í,µĽÖÂłĆÁżµÄąĚĚĺÖĘÁżĆ«Đˇ,ČÜÖʵÄÎďÖʵÄÁżĆ«Đˇ,ČÜҺŨ¶ČĆ«µÍ,ąĘѡŁ»
˘ÚËůÓõÄíŔÂëÉúĐâ,µĽÖÂłĆÁżµÄąĚĚĺÖĘÁżĆ«´ó,ČÜÖʵÄÎďÖʵÄÁżĆ«´ó,ČÜҺŨ¶ČĆ«¸ß,ąĘ˛»ŃˇŁ»
˘ŰČÜҺתŇƵ˝ČÝÁżĆżşó,δ˝řĐĐĎ´µÓ˛Ů×÷,µĽÖ²ż·ÖČÜÖĘËđşÄ,ČÜÖʵÄÎďÖʵÄÁżĆ«Đˇ,ČÜҺŨ¶ČĆ«µÍ,ąĘѡŁ»
˘ÜתŇĆČÜҺǰČÝÁżĆżÄÚÓĐÉŮÁżŐôÁóË®,¶ÔČÜÖʵÄÎďÖʵÄÁżşÍČÜŇşĚĺ»ý¶Ľ˛»˛úÉúÓ°Ďě,ČÜҺŨ¶Č˛»±ä,ąĘ˛»ŃˇŁ»
˘Ý¶¨ČÝʱ,ŃöĘÓČÝÁżĆżµÄżĚ¶ČĎß,µĽÖÂČÜŇşĚĺ»ýĆ«´ó,ČÜҺŨ¶ČĆ«µÍ,ąĘѡŁ»
˘ŢÔÚÉŐ±ÖĐČÜ˝âNaOHşó,Á˘Ľ´˝«ËůµĂČÜҺעČëČÝÁżĆżÖĐ,ŔäČ´şó,ČÜŇşĚĺ»ýƫС,ČÜҺŨ¶ČĆ«¸ß,ąĘ˛»ŃˇŁ»
˘ß¶¨ČÝşóҡÔČ,·˘ĎÖŇşĂ潵µÍ,ÓÖ˛ąĽÓÉŮÁżË®,ÖŘĐ´ﵽżĚ¶ČĎß,µĽÖÂČÜŇşĚĺ»ýĆ«´ó,ČÜҺŨ¶ČĆ«µÍ,ąĘѡŁ»
˘ŕÓĂÁżÍ˛ÁżČˇŇ»¶¨ÁżĹ¨ÁňËáĹäÖĆϡÁňËᣬתŇƵ˝ÉŐ±şóŁ¬ÓĂÉŮÁżË®Ď´µÓÁżÍ˛Ł¬ÔěłÉËůȡÁňËáČÜÖʵÄÁżÔö´óŁ¬ČÜҺŨ¶ČĆ«¸ß,ąĘ˛»ŃˇŁ»·űşĎĚâŇâµÄѡĎî˘Ů˘Ű˘Ý˘ßˇŁ
ŐýȷѡĎî˘Ů˘Ű˘Ý˘ßˇŁ

ˇľĚâÄżˇżĎÂÁĐŔë×Ó·˝łĚĘ˝µÄĘéĐ´Ľ°ĆŔĽŰľůşĎŔíµÄĘÇ( )
ѡĎî | Ŕë×Ó·˝łĚĘ˝ | ĆŔĽŰ |
A | ˝«2 mol Cl2ͨČ뺬1 mol FeI2µÄČÜŇşÖĐŁş 2Fe2Ł«Ł«2IŁŁ«2Cl2===2Fe3Ł«Ł«4ClŁŁ«I2 | ŐýČ·Ł»Cl2ąýÁżŁ¬żÉ˝«Fe2Ł«ˇ˘IŁľůŃő»Ż |
B | Ba(HCO3)2ČÜŇşÓë×ăÁżµÄNaOHČÜŇş·´Ó¦Łş Ba2Ł«Ł«HCO3-Ł«OHŁ===BaCO3ˇýŁ«H2O | ŐýČ·Ł»ËáĘ˝ŃÎÓëĽî·´Ó¦ÉúłÉŐýŃÎşÍË® |
C | ąýÁżSO2ͨČëNaClOČÜŇşÖĐŁş SO2Ł«H2OŁ«ClOŁ===HClOŁ«HSO3- | ŐýČ·Ł»ËµĂ÷ËáĐÔŁşH2SO3ÇżÓÚHClO |
D | 1 mol/LµÄNaAlO2ČÜŇşşÍ2.5 mol/LµÄHClČÜŇşµČĚĺ»ý»ěşĎŁş 2AlO2-Ł«5HŁ«===Al3Ł«Ł«Al(OH)3ˇýŁ«H2O | ŐýČ·Ł»µÚŇ»˛˝·´Ó¦şÍµÚ¶ţ˛˝·´Ó¦ĎűşÄµÄHŁ«µÄÎďÖʵÄÁżÖ®±ČÎŞ2ˇĂ3 |
A. A B. B C. C D. D
ˇľĚâÄżˇżSO2ÓëĆŻ·Űľ«ĘÇłŁÓõÄĆŻ°×ĽÁˇŁÄłĐËȤС×é¶ÔËüĂǵÄĆŻ°×ÔŔí˝řĐĐĚ˝ľżˇŁąýłĚČçĎÂŁş
I. Ě˝ľżSO2µÄĆŻ°×ĐÔ
ʵŃéŇ»Łş˝«SO2·Ö±đͨČë0.1%Ć·şěË®ČÜŇşşÍ0.1%Ć·şěŇŇ´ĽČÜŇşÖĐŁ¬ąŰ˛ěµ˝Ç°ŐßÍĘÉ«¶řşóŐ߲»ÍĘÉ«ˇŁ
ʵŃé¶ţŁş
| ĘÔąÜÖеÄŇşĚĺ | ĎÖĎó |
a. 0.1 mol/L SO2ČÜŇş(pH=2) | ČÜŇşÖ𽥱äÇłŁ¬ÔĽ90sşóÍęČ«ÍĘÉ« | |
b. 0.1 mol/L NaHS03ČÜŇş(pH=5) | ČÜŇşÁ˘Ľ´±äÇłŁ¬ÔĽ15sşóÍęČ«ÍĘÉ« | |
c. 0.1 mol/L Na2SO3ČÜŇş( pH=10) | ČÜŇşÁ˘Ľ´ÍĘÉ« | |
d. pH=10NaOHČÜŇş | şěÉ«ČÜŇş˛»±äÉ« | |
e. pH=2H2SO4ČÜŇş | şěÉ«ČÜŇş˛»±äÉ« |
(1)ʵŃédµÄÄżµÄĘÇ____________ˇŁ
(2)ÓÉʵŃéŇ»ˇ˘¶ţżÉÖŞŁş¸ĂʵŃéĚőĽţĎÂŁ¬SO2ʹƷşěČÜŇşÍĘɫʱĆđÖ÷ŇŞ×÷ÓõÄ΢ÁŁĘÇ____________ˇŁ
(3)ŇŃÖŞS02ʹƷşěČÜŇşÍĘÉ«ąýłĚĘÇżÉÄćµÄˇŁĐËȤС×éĽĚĐřĘÔŃ飺ĎňaʵŃéşóµÄÎŢÉ«ČÜŇşÖеÎČëBa(OH)2ČÜŇşÖÁpH=10ʱŁ¬ÉúłÉ°×É«łÁµíŁ¬ČÜŇş±äşěˇŁ
ÇëÓĂŔë×Ó·˝łĚĘ˝şÍ±ŘŇŞµÄÎÄ×Ö˵Ă÷łöĎÖÉĎĘöĎÖĎóµÄÔŇň____________ˇŁ
II. Ě˝ľżSO2ÓëĆŻ·Űľ«µÄ·´Ó¦
ʵŃéČýŁş
˛Ů×÷ | ĎÖĎó |
| i. ŇşĂćÉĎ·˝łöĎÖ°×ÎíŁ» ii. ÉԺ󣬳öĎÖ»ë×ÇŁ¬ČÜŇş±äÎŞ»ĆÂĚÉ«Ł» iii. ÉԺ󣬲úÉú´óÁż°×É«łÁµíŁ¬»ĆÂĚÉ«ÍĘČĄ |
С×éͬѧ¶ÔÉĎĘöĎÖĎó˝řĐĐĚ˝ľżŁş
(1)ĎňË®ÖĐłÖĐřͨČëSO2Ł¬Î´ąŰ˛ěµ˝°×ÎíˇŁÍƲâĎÖĎóiµÄ°×ÎíÓÉHClСҺµÎĐγɣ¬˝řĐĐČçĎÂʵŃ飺
a. ÓĂĘŞČóµÄµâ»ŻĽŘµí·ŰĘÔÖ˝ĽěŃé°×ÎíŁ¬Îޱ仯Ł»
b. ÓĂËữµÄAgNO3ČÜŇşĽěŃé°×ÎíŁ¬˛úÉú°×É«łÁµíˇŁ
˘ŮʵŃéaÄżµÄĘÇ____________________ˇŁ
˘ÚÓÉʵŃéaˇ˘b˛»ÄÜĹжϰ×ÎíÖĐş¬ÓĐHClŁ¬ŔíÓÉĘÇ____________ˇŁ
(2)ĎÖĎóiiÖĐČÜŇş±äÎŞ»ĆÂĚÉ«µÄżÉÄÜÔŇňŁşËćČÜŇşËáĐÔµÄÔöÇżŁ¬ĆŻ·Űľ«µÄÓĐЧłÉ·ÖşÍCl-·˘Éú·´Ó¦ˇŁÍ¨ąý˝řŇ»˛˝ĘµŃéČ·ČĎÁËŐâÖÖżÉÄÜĐÔŁ¬ĆäʵŃé·˝°¸ĘÇ____________ˇŁ
(3)˝«AĆżÖĐ»ěşĎÎďąýÂˡ˘Ď´µÓŁ¬µĂµ˝łÁµíX
˘ŮĎňłÁµíxÖĐĽÓČëϡHClŁ¬ÎŢĂ÷ĎԱ仯ˇŁČˇÉϲăÇĺŇşŁ¬ĽÓČëBaCl2ČÜŇşŁ¬˛úÉú°×É«łÁµíˇŁÔňłÁµíXÖĐş¬ÓеÄÎďÖĘĘÇ____________ˇŁ
˘ÚÓĂŔë×Ó·˝łĚĘ˝˝âĘÍĎÖĎóiiiÖĐ»ĆÂĚÉ«ÍĘČĄµÄÔŇň________________ˇŁ