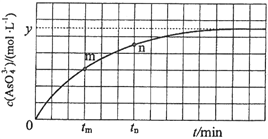
��Ŀ����
����Ŀ��һЩ��ˮ�����������������Ʒʱ��������ν�������ˮ����ʵ�飬������ˮ������ˮ���жԱȡ������װ�ò��봿��ˮ������ˮ��ͨ�������Ӻ���ˮ��ɫ�ޱ仯�����Ի�ɫ��������ˮȴð�����ݲ����ɻ���ɫ�����ɫ�ij���������������������ѡ��˵����ȷ���ǣ�������

A.ͨ��ʱ����Ƭ����������Ƭ������
B.���ɵ����ݡ�����ɫ�����ɫ�ij������ǵ���ֱ�ӻ��Ӳ���
C.ͨ���Աȣ�ȷʵ��˵������ˮ�к��д����������ж��к�������
D.��Ƭ�ĵ缫��Ӧ��O2+2H2O+4e-=4OH-
���𰸡�B
��������
����ˮȴð�����ݲ����ɻ���ɫ�����ɫ�ij�����˵��Feʧ���������������ӣ���ˮ��������������ӷ�Ӧ�����������������䱻�������ɻ���ɫ���ʻ�������������ɫ������
A. ������֪��ͨ��ʱ����Ƭ����������Ƭ�������������ⲻ����A����
B. ������ˮ�õ������ɵ����ݡ�����������ʧ���������������ӣ�Ȼ�����ɺ���ɫ�����ɫ�ij������ǵ���ֱ�ӻ��Ӳ���������⣬B��ȷ��
C. ͨ���Աȣ���˵������ˮ�к��д��������ӣ���һ��Ϊ�������ж��к������ʣ������ⲻ����C����
D. ��Ƭ���������缫��Ӧ��Fe-2e-=Fe2+�������ⲻ����D����
��ΪD��