��Ŀ����
����Ŀ�� �������ڲ�ͬ�ܼ����� NaOH ������ͬ���͵ķ�Ӧ�����ɲ�ͬ�ķ�Ӧ���ij ͬѧ��������������ʣ�����ͼʵ��װ�� ������̨���ƾ������� ��֤ȡ����Ӧ����ȥ ��Ӧ�IJ������һ�����̽����

ʵ������������Թ��м��� 5mL1mol/LNaOH ��Һ�� 5mL �����飬�� ʵ����� II�����Թ���ͼ�̶���ˮԡ���ȡ�
��1�� ��ˮԡ���ȶ���ֱ���þƾ��Ƽ��ȵ�ԭ���� ��
��2�� �۲쵽 ����ʱ�������������� NaOH ��Һ����ȫ ��Ӧ��
��3�� �������������Ҵ��Ľṹ�����õIJ����� ��
��4�� Ϊ֤���������� NaOH �Ҵ���Һ�з���������ȥ��Ӧ��������Ƶ�ʵ�鷽������ ��Ҫ������� ������ķ����� ��
����˵�������õ��Լ�����ʵ�������Ԥ�������ʵ����������
���𰸡�(9��)
��1��������е�ͣ��������������ʧ��2�֣�
��2���Թ�����Һ���úֲ㣨2�֣�
��3��������ס��˴Ź������ף�2�֣�
��4�����ɵ����壨1�֣������ɵ�������ͨ��ʢ��ˮ���Թܣ���ͨ��ʢ��KMnO4��Һ���Թܣ�KMnO4��Һ��ɫ(��ֱ��ͨ��������Ȼ�̼��Һ)��2�֣�
��������
�����������1��������е�ͣ��������ӷ�����ˮԡ�����Ⱦ��ȣ��������������ʧ���ʴ�Ϊ��ʹ�Թ����Ⱦ��ȣ��������������ʧ��
��2�������鲻����ˮ����ʼ��Һ�ֲ㣬���ɵIJ����Ҵ����廯�ƶ�������ˮ������Һ�ֲ���ʧ��������������NaOH��Һ����ȫ��Ӧ����Ӧ��ѧ����ʽΪ��CH3CH2Br+NaOH��CH3CH2OH+NaBr��
�ʴ�Ϊ���Թ�����Һ���úֲ㣻
��3���Ҵ��ĵ���ʽ��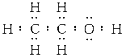 ���ṹʽ�ǣ�
���ṹʽ�ǣ�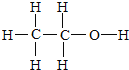 ���Ҵ����ӽṹ����������ԭ�ӣ����ǵı�Ϊ3��2��1�����ú˴Ź������ɼ�⣬Ҳ���ú������⣬
���Ҵ����ӽṹ����������ԭ�ӣ����ǵı�Ϊ3��2��1�����ú˴Ź������ɼ�⣬Ҳ���ú������⣬
�ʴ�Ϊ��������ס��˴Ź������ף�
��4�����������������ƵĴ���Һ�з�����ȥ��Ӧ����Ӧ��1��C��C-Br����CH3CH2Br+NaOH��
CH2=CH2��+NaBr+H2O��������ϩ��ֻ��֤����ϩ�Ĵ��ڼ���֤������������ȥ��Ӧ�������ǣ������ɵ�������ͨ��ʢ��ˮ���Թܣ���ͨ��ʢ������KMnO4��Һ���Թܣ�5CH2=CH2+12KMnO4+18H2O��12MnSO4+6K2SO4+10CO2��+28H2O������KMnO4��Һ��ɫ������ֱ��ͨ��������Ȼ�̼��Һ����ϩ���嵥�ʷ����ӳɣ�Br2+CH2=CH2��BrCH2-CH2Br�����Ȼ�̼��ɫ����
�ʴ�Ϊ�����ɵ����壻�����ɵ�������ͨ��ʢ��ˮ���Թܣ���ͨ��ʢ������KMnO4��Һ���Թܣ�����KMnO4��Һ��ɫ����ֱ��ͨ��������Ȼ�̼��Һ�����Ȼ�̼��ɫ����







