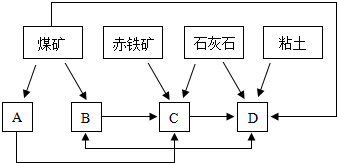
��Ŀ����
��ҵ����������ʱ�����ô�������Ӧ��SO2ת��ΪSO3��һ���ؼ����衣ѹǿ���¶ȶ�SO2ת���ʵ�Ӱ�����±�(ԭ�������ɷֵ��������Ϊ��SO2 7%��O2 11%��N2 82%)

(1)��֪SO2�������Ƿ��ȷ�Ӧ��������ñ��������ƶϴ˽��ۣ�________________________________�� (2)��400 �桫500 ��ʱ��SO2�Ĵ��������ó�ѹ�����Ǹ�ѹ����Ҫԭ���ǣ�_____________________________��
(3)ѡ�����˵Ĵ������Ƿ�������SO2��ת���ʣ�_______(��ǡ���)���Ƿ��������÷�Ӧ���ų���������______(��ǡ���)��
(4)Ϊ���SO3�����ʣ�ʵ����������_________����SO3��
(5)��֪��2SO2(g)��O2(g) = 2SO3(g)����H =��196.6 kJ/mol������ÿ����1���98%��������Ҫ��SO3����������SO2������ЩSO3���ų���������
(3)ѡ�����˵Ĵ������Ƿ�������SO2��ת���ʣ�_______(��ǡ���)���Ƿ��������÷�Ӧ���ų���������______(��ǡ���)��
(4)Ϊ���SO3�����ʣ�ʵ����������_________����SO3��
(5)��֪��2SO2(g)��O2(g) = 2SO3(g)����H =��196.6 kJ/mol������ÿ����1���98%��������Ҫ��SO3����������SO2������ЩSO3���ų���������
(1)ѹǿһ��ʱ���¶����ߣ�SO2ת�����½���˵�������������淴Ӧ���У���������ӦΪ���ȷ�Ӧ
(2)����ѹǿ�����SO2ת����������Ӱ�죬���������ӳɱ�
(3)��
(4)Ũ����
(2)����ѹǿ�����SO2ת����������Ӱ�죬���������ӳɱ�
(3)��
(4)Ũ����
(5)

��ã�m(SO3) = 8.0��109g = 8.0��103t��Q = 9.83��109 kJ��

��ϰ��ϵ�д�
�����Ŀ