��Ŀ����
��15�֣�����A��B�����л��������֪���� B���ӿ��Կ���A������һ����ԭ�ӱ�����ȡ�����õ��������ǵķ�������Ԫ�ص�����������ͬ����Է���������������170��A��̼Ԫ�ص���������Ϊ72.00���������Ƕ���һȡ�����������ж���������������һ���Ȼ���
�밴Ҫ����գ�
��1��A��B�ķ�������Ԫ�ص������������� ��B�ķ���ʽΪ ��
��2��A�Ľṹ��ʽ�� ��
��3��C��A��Ϊͬ���칹�壬������Ҳû�м��������������������ڶ�λ��ȡ�������ܷ���������Ӧ��Ҳ����һ�������·�����ȥ��Ӧ����C��������Һ��Ӧ�Ļ�ѧ����ʽ�� ��
��4��B��һ��ͬ���칹��D��ij����������������Ӧ���ɵ�Ψһһ��һ����ȡ����������ڱ����ϣ�����D�Ľṹ��ʽ�� ��
�밴Ҫ����գ�
��1��A��B�ķ�������Ԫ�ص������������� ��B�ķ���ʽΪ ��
��2��A�Ľṹ��ʽ�� ��
��3��C��A��Ϊͬ���칹�壬������Ҳû�м��������������������ڶ�λ��ȡ�������ܷ���������Ӧ��Ҳ����һ�������·�����ȥ��Ӧ����C��������Һ��Ӧ�Ļ�ѧ����ʽ�� ��
��4��B��һ��ͬ���칹��D��ij����������������Ӧ���ɵ�Ψһһ��һ����ȡ����������ڱ����ϣ�����D�Ľṹ��ʽ�� ��
��1��6.67% C9H11NO2��2��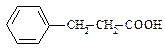
��3��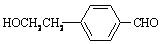 +2Ag(NH3)2OH
+2Ag(NH3)2OH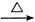
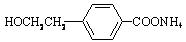 +2Ag+
+2Ag+
3NH3+H2O
��4��

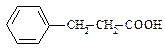
��3��
 +2Ag(NH3)2OH
+2Ag(NH3)2OH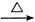
 +2Ag+
+2Ag+3NH3+H2O
��4��

��

��ϰ��ϵ�д�
�����Ŀ


 �ش��������⣺
�ش��������⣺ ��
�� ��
��



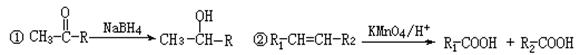
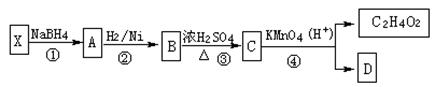
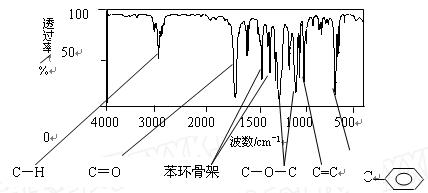
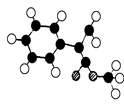
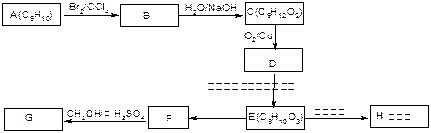
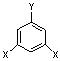 ��ʾ������X��Y����ΪH��
��ʾ������X��Y����ΪH��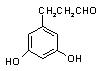 ��
��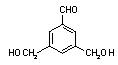 �� ��
�� ��