��Ŀ����
��1���Ӻ�ˮ�п���ȡ���ֻ���ԭ�ϣ������ǹ�ҵ�϶Ժ�ˮ�ļ����ۺ����õ�ʾ��ͼ��

��.��ˮ���ô����к�Ca2+��Mg2+��SO �� �����ʣ�Ϊ����Ч��ȥCa2+��Mg2+��SO
�� �����ʣ�Ϊ����Ч��ȥCa2+��Mg2+��SO ���������Լ��ĺ���˳��Ϊ___________��
���������Լ��ĺ���˳��Ϊ___________��
A���ȼ�NaOH�����Na2CO3���ټ�Ba2+�Լ�
B���ȼ�NaOH�����Ba2+�Լ����ټ�Na2CO3
C���ȼ�Ba2+�Լ������NaOH���ټ�Na2CO3
Ҫ��ȥSO �����������ӱ��Լ����ñ��Լ�������___________��
�����������ӱ��Լ����ñ��Լ�������___________��
A��Ba(OH)2 B�� Ba(NO3)2 C��BaCl2
��.д����Ca(OH)2��ȡ������þ�Ļ�ѧ����ʽ��_____________________��
�Ӿ���Ч��Ƕȿ����û������ij�ַӦѡ����__________________��
��2����������ȡ���������ͼ��
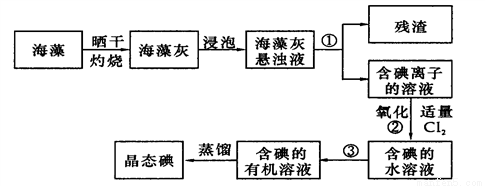
ָ����ȡ��Ĺ������й�ʵ��������ƣ���__________����__________��
д�����̢����йط�Ӧ�����ӷ���ʽ____________________________��
 ���ɶ���ܲ��¿�ֱͨ��Уϵ�д�
���ɶ���ܲ��¿�ֱͨ��Уϵ�д�����ʵ�顰�����������롰���ۡ���Ӧ��ϵ��ȷ���ǣ� ��
ѡ�� | ʵ����������� | ʵ����� |
A | ��ij��Һ�м����Ȼ�����Һ���а�ɫ�������ɣ��ټ������ữ���������ܽ� | ����Һ��һ������SO42�� |
B | ��ij��Һ��ͬʱ���뼸��KSCN��Һ���������Ƶ���ˮ����Һ��Ϊ��ɫ | ����Һ��һ������Fe2+ |
C | ��ij��Һ�м���ϡ���ᣬ������ʹ����ʯ��ˮ����ǵ���ɫ���� | ����Һ��һ������CO32�� |
D | ��ij��Һ����NaOH���ȣ������ܹ�ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���� | ����Һ��һ������NH4+ |
A. A B. B C. C D. D




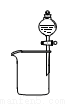 B. ��ȥCO2�е�HCl
B. ��ȥCO2�е�HCl 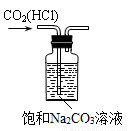
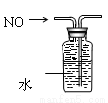
 D. ��ˮ���ռ�NO
D. ��ˮ���ռ�NO