��Ŀ����
����Ŀ��Ϊ�ⶨijͭ������Cux(OH)yClz��CuΪ��2�ۣ�x��y��zΪ������������ɣ��ֽ�������ʵ�飺ȡ����Cux(OH)yClz��������ȫ�ܽ���ϡ�����У���������Һ�ֳ����ȷݡ�������һ�ݵμ�����AgNO3��Һ�������ó������ˡ�ϴ�ӡ�����õ�����0.1435 g������һ���м���0.1000 mol��L1 EDTA(��֪Cu2+��EDTA��Ӧ�Ļ�ѧ��������Ϊ1��1)��ǡ����ȫ��Ӧʱ����EDTA 20mL����Cux(OH)yClz�Ļ�ѧʽ��y��ֵΪ�� ��
A.1B.2C.3D.4
���𰸡�C
��������
�����зݵμ�����AgNO3��Һ�������ó������ˡ�ϴ�ӡ�����õ�����0.1435g���ɴ˼���n(Cl-)������һ���м���0.1000mol��L-1EDTA����֪Cu2+��EDTA��Ӧ�Ļ�ѧ��������Ϊ1��1����ǡ����ȫ��Ӧʱ����EDTA 20mL���ɴ˼���n(Cu2+)��Ȼ���ϻ�ѧʽ��Ԫ�صĻ��ϼ۴�����Ϊ����㻯ѧʽ��y��ֵ��
�����зݵμ�����AgNO3��Һ�������ó������ˡ�ϴ�ӡ�����õ�����0.1435g���������Ȼ���������n(AgCl)= ![]() =0.001mol�������������ʵ���=0.001mol������һ���м���0.1000mol��L-1EDTA����֪Cu2+��EDTA��Ӧ�Ļ�ѧ��������Ϊ1��1����ǡ����ȫ��Ӧʱ����EDTA 20mL��n(Cu2+)=0.1000molL-1��0.02L=0.002mol��Cux(OH)yClz��n(Cu2+)��n(Cl-)=2��1����x=2���ɻ�ѧʽ��Ԫ�صĻ��ϼ۴�����Ϊ�㣺��+2����2+��-1����1+��-1����y=0�����y=3����ΪC��
=0.001mol�������������ʵ���=0.001mol������һ���м���0.1000mol��L-1EDTA����֪Cu2+��EDTA��Ӧ�Ļ�ѧ��������Ϊ1��1����ǡ����ȫ��Ӧʱ����EDTA 20mL��n(Cu2+)=0.1000molL-1��0.02L=0.002mol��Cux(OH)yClz��n(Cu2+)��n(Cl-)=2��1����x=2���ɻ�ѧʽ��Ԫ�صĻ��ϼ۴�����Ϊ�㣺��+2����2+��-1����1+��-1����y=0�����y=3����ΪC��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ�����ײ���һֱ�������о�����Ҫ���⣬��������Fe�۱�������г�ǿ�Ĵ��ԣ���Ч���Ե����������ʡ�
I��ʵ���Ҳ������ԭ���Ʊ�����Fe����������ͼ��ʾ��
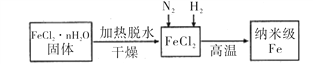
��1������Fe��ϡ���ᷴӦ�����ӷ���ʽΪ_______________________________��
��2����ν�FeCl2��nH2O���������ˮ�Ƶ���ˮFeCl2 _____________________________________(�ü�Ҫ��������)��
��3����������Fe�Ļ�ѧ����ʽΪ______________________________________��
II���������ϣ��ڲ�ͬ�¶��£�����Fe����ˮ������Ӧ�Ĺ�����ﲻͬ���¶ȵ���570��ʱ����FeO������570��ʱ����Fe3O4����ͬѧ����ͼ��װ����ʾ��������Fe����ˮ������Ӧ��ʵ�飬��ͬѧ��ͼ����ʾ��װ�ý�������Fe����ˮ�����ķ�Ӧ����֤���
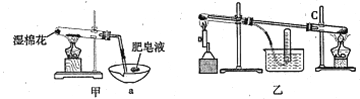
��4����װ��������Fe����ˮ������Ӧ�Ļ�ѧ����ʽ�� ______________________��
��5����װ��������a������Ϊ_______________________��
��6����ͬѧΪ̽��ʵ��������Թ��ڵĹ������ʳɷ֣�����������ʵ�飺
ʵ�鲽�� | ʵ����� | ʵ������ |
I | ����Ӧ��õ��ĺ�ɫ��ĩX(�ٶ�Ϊ���ȵ�)��ȡ������������һ�Թ��У������������ᣬ�� | ��ɫ��ĩ���ܽ⣬��Һ��dz��ɫ�����������ݲ��� |
II | ��ʵ��I�õ�����Һ�еμӼ���KSCN��Һ���� | ��Һû�г��ֺ�ɫ |
��������ʵ�飬��ͬѧ��Ϊ�������·�Ӧ�Ĺ������ΪFeO��
��ͬѧ��Ϊ��ͬѧ�Ľ��۲���ȷ������������______(�ü�Ҫ��������)��
��7����ͬѧ��ȡ5.60gFe�ۣ�����װ��Ӧһ��ʱ���ֹͣ���ȡ����Թ��ڵĹ��������ڸ���������ȴ�Ƶ�����Ϊ6.88g����ͬѧʵ���Ĺ������������������������Ϊ________(���������λ��Ч����)��