��Ŀ����
ijͬѧ�о������仯���������ʱ�������������ʵ�鷽����
����һ��2.7 g Al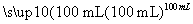 X��Һ
X��Һ Al(OH)3����
Al(OH)3����
��������2.7 g Al Y��Һ
Y��Һ Al(OH)3����
Al(OH)3����
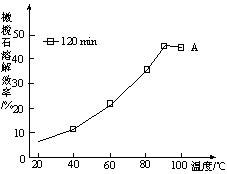
��֪��NaOH��Һ��ϡ�����Ũ�Ⱦ���3 mol��L��1����ͼ����X��Һ��Y��Һ�зֱ����NaOH��Һ��ϡ����ʱ�������������ʵ�����������������������Һ���֮��Ĺ�ϵ������˵����ȷ����(����)
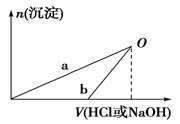
A����O��ʱ��������������Һ���������ʵ������
B��b���߱�ʾ������X��Һ�м���NaOH��Һ
C��X��Һ����ΪAlCl3��Y��Һ����ΪNaAlO2
D��a��b���߱�ʾ�ķ�Ӧ����������ԭ��Ӧ
A

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ
���ֶ�����Ԫ�ص�ԭ�Ӱ뾶����Ҫ���ϼ����±���
| Ԫ�ش��� | X | Y | Z | M | R |
| ԭ�Ӱ뾶/nm | 0.186 | 0.102 | 0.075 | 0.074 | 0.143 |
| ��Ҫ���ϼ� | ��1 | ��6 ��2 | ��5 ��3 | ��2 | ��3 |
��֪X�Ƕ�����������õĽ���������Rͬ���ڡ�������������ȷ���� ( )
A�����Ӱ뾶��С��R3��<X��< M2��
B��Y��M��ɵĻ������ǹ⻯ѧ������ԭ��֮һ
C����YM2ͨ��Ba(NO3)2��Һ���д�����ɫ��������
D��M�⻯����۷е����Z�⻯����۷е�
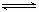 SO3��NO
SO3��NO 2MgCl2(aq) +SiO2 (s) + 2H2O(l) ��H =��49.04 kJ��mol-1
2MgCl2(aq) +SiO2 (s) + 2H2O(l) ��H =��49.04 kJ��mol-1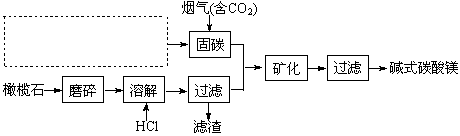
 ��2����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH(aq)+CO2 (g)=NaHCO3 (aq)���÷�Ӧ���Է����е�ԭ���� ��
��2����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH(aq)+CO2 (g)=NaHCO3 (aq)���÷�Ӧ���Է����е�ԭ���� ��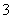 ��Na�� ��B��H����Br����SO
��Na�� ��B��H����Br����SO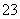 ��K��
��K��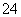 ��Br����OH����Fe2�� ��D��H����Br����SO
��Br����OH����Fe2�� ��D��H����Br����SO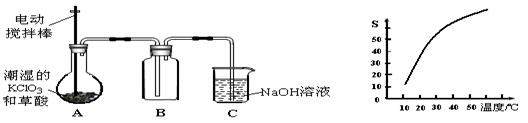
 ��SO
��SO ����������B��Na����Ba2����CO
����������B��Na����Ba2����CO ��NO
��NO