��Ŀ����
2008��5��12��14ʱ28�֣��ҹ��Ĵ�ʡ���Ӳ���Ǽ�������ݵ��봨�ط�����8.0�����������˼������Ա�����ͲƲ���ʧ�����ֹ����У�������Ա���й�עˮ�ʣ�ʱʱ��⡣�й���ԭ���˵���������
- A.���ˮ���д�Ⱥ��ָ��������ֹ���ú��³�����Ⱦ���ķ����ʼ�������
- B.��ֹ��ˮ��ʬ���ж�
- C.���ˮ�ĺ��������Ƿ��ܼ�ʱ��Ӧ��������
- D.����ˮ�dz�����Ⱦ���Ĵ���;��֮һ
C
����ˮ�ʼ����Ҫ��Ϊ�˴���֮�����ߡ�������Ⱦ�����ɸ��ֲ�ԭ�徭�����볦�����ɷ���ų���ԭ���һ�ּ�������������ˮԴ�ձ������ܵ��л���Ⱦ����Ⱥ�ձ鳬�����ұ������⣬ʬ��Ѹ�ٸ���������ʬ�������ʹ������Ⱦ���������У���������в������������������彡��
����ˮ�ʼ����Ҫ��Ϊ�˴���֮�����ߡ�������Ⱦ�����ɸ��ֲ�ԭ�徭�����볦�����ɷ���ų���ԭ���һ�ּ�������������ˮԴ�ձ������ܵ��л���Ⱦ����Ⱥ�ձ鳬�����ұ������⣬ʬ��Ѹ�ٸ���������ʬ�������ʹ������Ⱦ���������У���������в������������������彡��

��ϰ��ϵ�д�
 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�
�����Ŀ
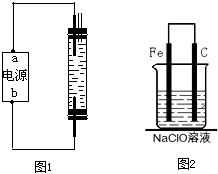 ��2010?��̨һģ��2008��5��12���ҹ��Ĵ��봨�����ش����Ϊ��ֹ�ڴ���֮���߲����У�ȫ�����������������˴����ĸ�������Һ����NaClO��Һ��ijУ̽����ѧϰС�������Һ�������ƣ�NaClO�����Ʊ������ʵȽ�����̽����
��2010?��̨һģ��2008��5��12���ҹ��Ĵ��봨�����ش����Ϊ��ֹ�ڴ���֮���߲����У�ȫ�����������������˴����ĸ�������Һ����NaClO��Һ��ijУ̽����ѧϰС�������Һ�������ƣ�NaClO�����Ʊ������ʵȽ�����̽���� �������ϣ���1��2007��11��26�գ��ҹ��״�����̽��̵�һ������ͼ��������������к��зḻ��3He���º����������̲��ŷḻ�Ľ��������Դ�ʹ����Ķ������衢����ȣ� ��2��2008��5��12���Ĵ�����ǿ�ҵ��𣮵��������ڵ������ʯ���ѡ������ѳ��ڻ������������������ͷų���������֪������ı���������ʯ�����������ʯ��Ҫ�ɷֶ��ǹ����Σ���������������ǣ�������
�������ϣ���1��2007��11��26�գ��ҹ��״�����̽��̵�һ������ͼ��������������к��зḻ��3He���º����������̲��ŷḻ�Ľ��������Դ�ʹ����Ķ������衢����ȣ� ��2��2008��5��12���Ĵ�����ǿ�ҵ��𣮵��������ڵ������ʯ���ѡ������ѳ��ڻ������������������ͷų���������֪������ı���������ʯ�����������ʯ��Ҫ�ɷֶ��ǹ����Σ���������������ǣ�������