��Ŀ����
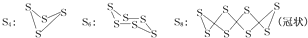
��֪��������ͨ����������S8(б����)����ʽ���ڣ���������״̬ʱ������S2��S4��S6��S8�ȶ���ͬ�������壬����S4��S6��S8�������ƵĽṹ�ص㣬��ṹ����ͼ��ʾ��

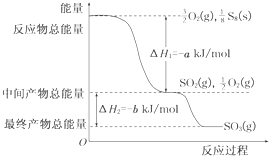
��һ�������£�S8(s)��O2(g)������Ӧ����ת��ΪSO2(g)��SO3(g)����Ӧ���̺�������ϵ������ͼ��ʾ(ͼ�еĦ�H��ʾ����1 mol������������)��

(1)д����ʾS8ȼ���ȵ��Ȼ�ѧ����ʽ_____________________��
(2)д��SO3�ֽ�����SO2��O2���Ȼ�ѧ����ʽ_________________________��
(3)����֪SO2���������ļ���Ϊd kJ��mol-1��O2���������ļ���Ϊe kJ��mol-1����S8������������ļ���Ϊ___________��
(1)S8(s)+8O2(g)==8SO2(g) ��H=-8a kJ�Mmol
(2)2SO3(g) 2SO2(g)+O2(g) ��H=+2bkJ�Mmol
2SO2(g)+O2(g) ��H=+2bkJ�Mmol
(3)(2d-a-e) kJ�Mmol
��������
�����������ͼ�ɿ�������1mol�Ķ�������ų�����ΪaKJ�Mmol������S8ȼ�յ��Ȼ�ѧ����ʽΪ�� S8��s��+8O2(g)=8SO2(g ) ; ��H=-8aKJ�Mmol (2) SO2��O2�� �����ߣ�SO3�����͡�SO3�ֽ�����SO2��O2�������ֽ�2���Ȼ�ѧ����ʽΪ��2SO3(g) 2SO2(g)+O2(g) ��H=+2b kJ�Mmol.���ݷ�Ӧ�Ⱦ��Ƕ��Ѿɼ����յ��������γ��»�ѧ�����ͷŵ������IJ�ֵ������S��S��ΪX����8X+8e-16d=-8a ,���X=��2d-a-e��kJ�Mmol.
2SO2(g)+O2(g) ��H=+2b kJ�Mmol.���ݷ�Ӧ�Ⱦ��Ƕ��Ѿɼ����յ��������γ��»�ѧ�����ͷŵ������IJ�ֵ������S��S��ΪX����8X+8e-16d=-8a ,���X=��2d-a-e��kJ�Mmol.
���㣺���黯ѧ���뷴Ӧ�ȼ���Ӧ���뷴Ӧ��������������ϵ��֪ʶ��