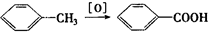��Ŀ����
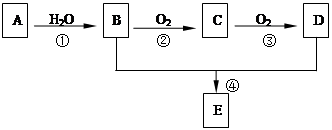
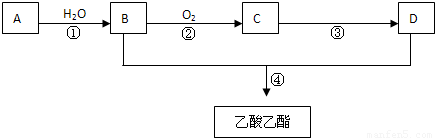
��֪���л���A�IJ���������������һ�����ҵ�ʯ�ͻ�����չˮƽ������AΪ��Ҫԭ�Ϻϳ�������������ϳ�·������ͼ��ʾ��

��1��A�����й����ŵ�������
��2����Ӧ�ڵĻ�ѧ����ʽ��
��Ӧ�ܵĻ�ѧ����ʽ��
��3��ijͬѧ����ͼ��ʾ��ʵ��װ����ȡ��������������ʵ��������Թܼ����ϲ�Ϊ���ġ�������ˮ����״Һ�壮
��ʵ�鿪ʼʱ���Թܼĵ��ܲ�����Һ���µ�ԭ����
����������Թܼ��и���״Һ����Ҫ�õ���������
a��©�� b����Һ©�� c������©��
��ʵ������������Թܼף�������ɫ�������ɣ�����Ҫԭ����

��1��A�����й����ŵ�������
̼̼˫��
̼̼˫��
����Ӧ�ٵĻ�ѧ��Ӧ�������ӳ�
�ӳ�
��Ӧ����2����Ӧ�ڵĻ�ѧ����ʽ��
2CH3CH2OH+O2
2CH3CHO+2H2O
| ��Cu |
2CH3CH2OH+O2
2CH3CHO+2H2O
��| ��Cu |
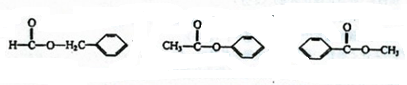
��Ӧ�ܵĻ�ѧ����ʽ��
CH3COOH+C2H5OH
CH3COOC2H5+H2O
| ŨH2SO4 |
| �� |
CH3COOH+C2H5OH
CH3COOC2H5+H2O
��| ŨH2SO4 |
| �� |

��3��ijͬѧ����ͼ��ʾ��ʵ��װ����ȡ��������������ʵ��������Թܼ����ϲ�Ϊ���ġ�������ˮ����״Һ�壮
��ʵ�鿪ʼʱ���Թܼĵ��ܲ�����Һ���µ�ԭ����
��ֹ��Һ����
��ֹ��Һ����
������������Թܼ��и���״Һ����Ҫ�õ���������
b
b
������ţ���a��©�� b����Һ©�� c������©��
��ʵ������������Թܼף�������ɫ�������ɣ�����Ҫԭ����
����ķе�ͣ�����ʱ��������������Թܼף���ʱ��������̼���ƽӴ���������Ӧ2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2������������
����ķе�ͣ�����ʱ��������������Թܼף���ʱ��������̼���ƽӴ���������Ӧ2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2������������
����ϻ�ѧ����ʽ�ش𣩣��������л���A�IJ���������������һ�����ҵ�ʯ�ͻ�����չˮƽ����A����ϩ����ϩ��ˮ��Ӧ�����Ҵ�����B���Ҵ����Ҵ���������������C��C��Ӧ����D��D���Ҵ���Ӧ����������������D�����ᣬC����ȩ��
����⣺�л���A�IJ���������������һ�����ҵ�ʯ�ͻ�����չˮƽ����A����ϩ����ϩ��ˮ��Ӧ�����Ҵ�����B���Ҵ����Ҵ���������������C��C��Ӧ����D��D���Ҵ���Ӧ����������������D�����ᣬC����ȩ��
��1��A��ϩ����A�еĹ�������̼̼˫������ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ������Է�Ӧ�����Ǽӳɣ��ʴ�Ϊ��̼̼˫�����ӳɣ�
��2����ͭ�����������������£��Ҵ�����������������ȩ��ˮ����Ӧ����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O����Ũ���������������������£�������Ҵ�����������Ӧ����������������Ӧ����ʽΪCH3COOH+C2H5OH
CH3COOC2H5+H2O��
�ʴ�Ϊ��2CH3CH2OH+O2
2CH3CHO+2H2O��CH3COOH+C2H5OH
CH3COOC2H5+H2O��
��3���������ﺬ���Ҵ������ᣬ���߶�������ˮ�������ò��뵽Һ�����£���������������ʴ�Ϊ����ֹ��Һ������
�����ɵ��������������ڱ���̼������Һ��Ӧ�÷�Һ�ķ������룬�ʴ�Ϊ��b��
�������ӷ����������ԣ�����̼���Ʒ���2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2�����������ݣ�
�ʴ�Ϊ������ķе�ͣ�����ʱ��������������Թܼף���ʱ��������̼���ƽӴ���������Ӧ2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2�����������ݣ�
��1��A��ϩ����A�еĹ�������̼̼˫������ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ������Է�Ӧ�����Ǽӳɣ��ʴ�Ϊ��̼̼˫�����ӳɣ�
��2����ͭ�����������������£��Ҵ�����������������ȩ��ˮ����Ӧ����ʽΪ2CH3CH2OH+O2
| ��Cu |
| ŨH2SO4 |
| �� |
�ʴ�Ϊ��2CH3CH2OH+O2
| ��Cu |
| ŨH2SO4 |
| �� |
��3���������ﺬ���Ҵ������ᣬ���߶�������ˮ�������ò��뵽Һ�����£���������������ʴ�Ϊ����ֹ��Һ������
�����ɵ��������������ڱ���̼������Һ��Ӧ�÷�Һ�ķ������룬�ʴ�Ϊ��b��
�������ӷ����������ԣ�����̼���Ʒ���2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2�����������ݣ�
�ʴ�Ϊ������ķе�ͣ�����ʱ��������������Թܼף���ʱ��������̼���ƽӴ���������Ӧ2CH3COOH+Na2CO3=2CH3COONa+H2O+CO2�����������ݣ�
���������⿼���л�����ƶϣ�������ѧ������������ʵ�������Ŀ��飬ע����ճ����л���Ľṹ�����ʣ���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
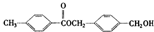 ����A�������ɷ���ͼʾ�е�һϵ�з�Ӧ������K�ķ���ʽΪC12H14O4��L��K��Ϊͬ���칹�壬F�IJ�������Ϊ����һ������ʯ�ͻ�����չˮƽ�ı�־����ͼ��[O]��ʾ������
����A�������ɷ���ͼʾ�е�һϵ�з�Ӧ������K�ķ���ʽΪC12H14O4��L��K��Ϊͬ���칹�壬F�IJ�������Ϊ����һ������ʯ�ͻ�����չˮƽ�ı�־����ͼ��[O]��ʾ������