��Ŀ����
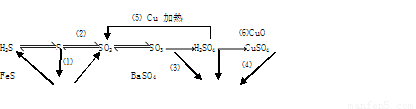
(16��)���仯����������ת����ϵ
(1)����������ԭ��Ӧ���� (�����)
(2)д��(5)�ķ�Ӧ����ʽ ��
˵��Ũ������� ����ȡCuSO4�� �������(�����)��
(3)SO2����ɿ�����Ⱦ���γ��������Ҫ���ʡ�SO2��ˮ�Ĵ�������������Ӧ�������ᣬ��Ӧ����ʽΪ ��
��֤��������ķ����� ��
(4)ij��Һ�к���Cl-��SO42-�����ܺ���Na+��Fe2+������һ�֡�
����֤Cl-��SO42-�ķ�����
A.�ȼ�BaCl2��Һ���ȳ������ټ�AgNO3��Һ
B.�ȼ�AgNO3��Һ���ȳ������ټ�BaCl2��Һ
C.�ȼ�Ba(NO3)2��Һ���ȳ������ټ�AgNO3��Һ
����֤Na+��Fe2+��ķ����� ��
(16��)
(1) (1)(2)(5) (2��)
(2)2H2SO4(Ũ)+Cu CuSO4+SO2��+2H2O�� (2��)
CuSO4+SO2��+2H2O�� (2��)
ǿ�������� (6) (��1��=2��)
(3) 2SO2+O2+2H2O===2H2SO4 (2��)
��ij����ͨ��ʢƷ����Һ���Թ�����Ʒ����ɫ���ټ������ֳ��ֺ�ɫ��֤����SO2�� (3��)
(4)�� C (2��)
������ҺΪ��ɫ������Һ��ֻ��Na+������Һ����ɫ��һ����Fe2+��������ɫ��Ӧ����֤��Na+�Ĵ��ڡ�(����������Ҳ����) (3��)
����

 ������������ϵ�д�
������������ϵ�д�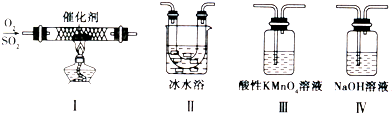
 ==6SO2��Fe3O4����������Ϊ ������3 mol FeS2�μӷ�Ӧ��
==6SO2��Fe3O4����������Ϊ ������3 mol FeS2�μӷ�Ӧ�� ��3�����������ƣ�������Ҳ��������ˮ������ԭ��Ϊ
�������ӷ��̱�ʾ��
��3�����������ƣ�������Ҳ��������ˮ������ԭ��Ϊ
�������ӷ��̱�ʾ��