��Ŀ����
��22�֣�2SO2(g)+O2(g) 2SO3(g)��Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2(g)����Ϊ1mol SO3�Ħ�H=��99kJ��mol��1����ش��������⣺
��1��ͼ��A��C�ֱ��ʾ �� ��E�Ĵ�С�Ը÷�Ӧ�ķ�Ӧ������Ӱ�죿 ���÷�Ӧͨ����V2O5����������V2O5��ʹͼ��B�������ǽ��ͣ� �������� ��
��2��ͼ�С�H= KJ��mol��1��
��3��V2O5�Ĵ�ѭ����������Ϊ��V2O5����SO2ʱ����������ԭΪ�ļ۷�������ļ۷��������ٱ�����������д���ô�ѭ�������Ļ�ѧ����ʽ ��
��4����2�֣������Ӧ���ʦԣ�SO2��Ϊ0��05 mol��L��1��min��1,��ԣ�O2��= mol��L��1��min��1��
��(SO3)= mol��L��1��min��1��
��5����2�֣���֪�������ȼ����Ϊ296 KJ��mol��1��������S(s)����3 molSO3(g)�ġ�H ��Ҫ�������̣���
��6������ȼ�ϵ�� ��KOH���������Һ��
������Ӧ����ʽ�ǣ�
������Ӧ����ʽ�ǣ�
�ܷ�Ӧ����ʽ�ǣ�
��7����2�֣������£���pH ��Ϊ5��H2SO4��A12(SO4)3��Һ����ˮ�������c(H+)�ֱ�Ϊc1 ��c2����c1��c2= ��
��8����2�֣�Ũ��Ϊ0.5 mol/L���������Ũ�ȵİ�ˮ��Һ��Ӧ��ʹ��Һ�����ԣ�����ǰ���V��________V��(����ڡ�����С�ڡ����ڡ�)
(9)��2�֣�ȡ10 mL��Һ0.5 mol/L�����ᣬ��ˮϡ�͵�500 mL�����ʱ��Һ����ˮ�������c(H��)��________��
��22�֣���1����Ӧ������ ���������� �� ���� ��Ϊ�����ı��˷�Ӧ������ʹ���E���� ��2����2�֣���198
(3) ��2�֣�SO2+V2O5=SO3+2VO2 4VO2+O2=2V2O5
(4)��2�֣�0��025 0��05
(5) ��2�֣�S(s)+O2(g)=2SO2(g) ��H1=��296KJ��mol��1 ,
SO2(g)+1/2O2(g) SO3(g)��H2=��99 KJ��mol��1
3 S(s)+9/2O2(g)=3SO3(g) ��H=3(��H1+��H2)=��1185 KJ��mol��1
��6��������CH4+10OH- ��8e-=CO32-+7H2O ������O2��2H2O+4e-=4OH-
�ܷ�Ӧ��CH4��2 O2+2OH-=CO32-+3H2O
��7����2�֣�1��104 ��8����2�֣�С�ڣ�9����2�֣�10-12
����:

 2SO3(g)��Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2(g)����Ϊ1mol�Ħ�H=��99kJ��mol��1����ش��������⣺
2SO3(g)��Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2(g)����Ϊ1mol�Ħ�H=��99kJ��mol��1����ش��������⣺
 CO(g)+3H2(g) �SH1="+206.1" kJ/mol
CO(g)+3H2(g) �SH1="+206.1" kJ/mol �����ʵ���Ũ���淴Ӧʱ��ı仯��ͼ1��ʾ��10minʱ���ı��������������� ��
�����ʵ���Ũ���淴Ӧʱ��ı仯��ͼ1��ʾ��10minʱ���ı��������������� ��
 ��ʹ�ס�����������ʼ�ݻ���ȡ�����ͬ�¶��·�����Ӧ�ڣ���ά�ַ�Ӧ�������¶Ȳ��䡣��֪��������
��ʹ�ס�����������ʼ�ݻ���ȡ�����ͬ�¶��·�����Ӧ�ڣ���ά�ַ�Ӧ�������¶Ȳ��䡣��֪��������
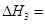 ��800��ʱ����Ӧ�۵Ļ�ѧƽ�ⳣ��K=1.0��ijʱ�̲�ø��¶��µ��ܱ������и����ʵ����ʵ������±���
��800��ʱ����Ӧ�۵Ļ�ѧƽ�ⳣ��K=1.0��ijʱ�̲�ø��¶��µ��ܱ������и����ʵ����ʵ������±���
 (��)
(��) (��) b��
(��) b�� 2SO3(g)����H=��96.56 kJ?mol-1���������ڷ�Ӧ�����б���ѹǿ���䣬����������������䣬������ά�־��ȣ����������Խ�����ѧƽ�⡣
2SO3(g)����H=��96.56 kJ?mol-1���������ڷ�Ӧ�����б���ѹǿ���䣬����������������䣬������ά�־��ȣ����������Խ�����ѧƽ�⡣