��Ŀ����
ͨ�����·�Ӧ���ɻ�ȡH2�������й�˵����ȷ���ǣ� ��
��̫������ֽ�ˮ���⣺2H2O(l)��2H2��g��+O2��g����H1=+571.6kJ��mol�C1
�ڽ�̿��ˮ��Ӧ���⣺C��s��+H2O(g)��CO��g��+H2��g����H2=+131.3kJ��mol�C1
�ۼ�����ˮ��Ӧ���⣺CH4��g��+H2O(g)��CO��g��+3H2��g����H3=+206.1kJ��mol�C1
A����Ӧ���е���ת��Ϊ��ѧ��
B����Ӧ��Ϊ���ȷ�Ӧ
C����Ӧ��ʹ�ô�������H3��С
D����ӦCH4��g����C��s��+2H2��g���Ħ�H =+74.8kJ��mol�C1
dz��ɫ����������茶���[����Ī���Σ���NH4)2SO4��FeSO4��6H2O]���̷���FeSO4�q7H2O)���ȶ��������ڶ���������Ī���ε�һ��ʵ�����Ʒ����£�
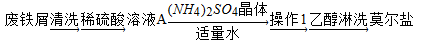
��1�������м�м���ϡ�����������м��ȫ�ܽ����ʣ������ʱ�ͽ��й��ˣ���Ŀ����________��
��2��0.10mol��L-1Ī������Һ������Ũ���ɴ�С��˳��Ϊ_________��
��3�������£���0.1 mol�qL-1 (NH4)2SO4��Һ��pH=5,����NH3�qH2O�ĵ���ƽ�ⳣ��Kb=___________
��4��Ϊ�˲ⶨ��Ʒ�Ĵ��ȣ���ȡag��Ʒ����ˮ�����Ƴ�500mL��Һ����Ũ��Ϊc mol�qL-1������KMnO4��Һ�ζ���ÿ����ȡ����Һ�����Ϊ25.00mL,ʵ������¼���£�
ʵ�����. | ��һ�� | �ڶ��� | ������ |
���ĸ��������Һ���/mL | .25.52, | 25.02 | 24.98 |
�ζ������з�����Ӧ�����ӷ���ʽΪ��_______________���ζ��յ��������____________________��
ͨ��ʵ�����ݼ���ĸò�Ʒ����Ϊ_________(����ĸac��)���ϱ��е�һ��ʵ���м�¼�������Դ��ں����Σ���ԭ�������____________(�����)��
A��ʵ�����ʱ���ӿ̶��߶�ȡ�ζ��յ�ʱ���Ը��������Һ�����
B���ζ�ǰ�ζ��ܼ��������ݣ��ζ�����������
C����һ�εζ��õ���ƿ�ô�װҺ��ϴ����������δ��ϴ
C����һ�εζ��õ���ƿ�ô�װҺ��ϴ����������δ��ϴ
D�������Ը�����ر�Һ����ʱ��������в��ֱ��ʣ�Ũ�Ƚ���


 l�������ڷ�Ӧ�еõ�1 mol����
l�������ڷ�Ӧ�еõ�1 mol���� CuSO4��A����2H2O
CuSO4��A����2H2O ���������� e������������
���������� e������������
 ��
�� ��
��