��Ŀ����
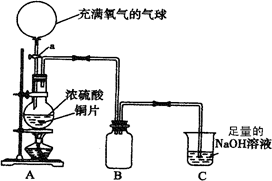
(18��)ij��ѧ��һ������ȤС��Ϊ̽��ͭ������ķ�Ӧ������������ͼ��ʾװ�ý����й�ʵ�顣
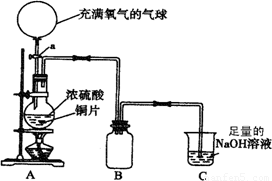
��1���ȹرջ���a����6.4gͭƬ��12 mLijŨ�ȵ�Ũ�������Բ����ƿ�й�������Ӧ��ϣ�������ƿ�л���ͭƬʣ�ࡣ�ٴ���a���������е�������������Բ����ƿ�����ͭƬ��ȫ��ʧ��
��д��������������ƿ�ڷ����Ļ�ѧ����ʽ���رջ���a ��
����a ��
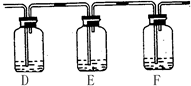
��B�������ռ�ʵ���в����������װ�ã�������δ��ȫ��������ͼ�аѵ��ܲ���������
��ʵ�������װ��C�е���Һ�п��ܺ��е������� ��
��2����С���ͬѧ�ԡ���μ���SO2�л�������CO2���������ܸ���Ȥ������A��ͭƬ����ľ̿�ۣ�����A��B֮������������װ�ã�
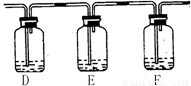
�Լ���a. NaOH��Һ b. Ʒ����Һ c. ����KMnO4��Һ d. Ca(OH)2��Һ
��ش�
�ٸ�ͬѧ��ʵ��װ��A�з����Ļ�ѧ����ʽ ��
����Ҫ�ﵽ��Ŀ�ģ������ڣ�(�����ṩ�Լ����)
D��� ��E��� ��F��� ��
��3����ʵ֤ʵ���ڣ�1����ͭƬ��ȫ��ʧ����������ʣ�࣬��ͬѧ���ⶨ��������ʵ���Ũ�ȣ����跴Ӧǰ����Һ����仯���Բ��ƣ�����Ӧ����Һ�м��뺬����a mol��NaOH��Һ�պ�ʹ��Һ��Cu2+ȫ���������ݴˣ����������������ʵ���Ũ�������ܣ���д���������ʵ���Ũ�ȵı���ʽ�� mol/L(�ú�a�Ĵ���ʽ��������ܣ��ÿղ���)��
��1����Cu+2H2SO4(Ũ)
 CuSO4+SO2+2H2O����2�֣�
CuSO4+SO2+2H2O����2�֣�
2Cu+O2+2H2SO4 2CuSO4+2H2O
��2�֣�
2CuSO4+2H2O
��2�֣�
(��ֲ�д�ɣ�2Cu+O2=2CuO��CuO+H2SO4=CuSO4+H2O��Ҳ��)
��
 (2��)
(2��)
��NaOH Na2SO3 Na2SO4 ��3�֣�
��2����C+2H2SO4��Ũ��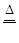 CO2+2SO2+2H2O
��3�֣�
CO2+2SO2+2H2O
��3�֣�
�� c b d ��3�֣�
��3�� ��3�֣������������ʽ���ɣ�
��3�֣������������ʽ���ɣ�
��������

 ��
�� ��NaOH��Һ�պ�ʹ��Һ��Cu2+ȫ���������ݴˣ����������������ʵ���Ũ�������ܣ���д���������ʵ���Ũ�ȵı���ʽ�� mol/L(
��NaOH��Һ�պ�ʹ��Һ��Cu2+ȫ���������ݴˣ����������������ʵ���Ũ�������ܣ���д���������ʵ���Ũ�ȵı���ʽ�� mol/L( �ú�a�Ĵ���ʽ��������ܣ��ÿղ���)��
�ú�a�Ĵ���ʽ��������ܣ��ÿղ���)��