��Ŀ����
16������ϩ����ͨ�����з����ϳɣ�
��1��A��B�ķ�Ӧ�ķ�Ӧ������ȡ����Ӧ���÷�Ӧ����������һ���л����ṹ��ʽΪCH3COOH��
��2��C�к��������ŵ��������ǻ���
��3���������D������C��ʵ�鷽����ȡ���������������������壬˵������C
��4��B�ж���ͬ���칹�壮E��B�ķ�����ͬ���칹�壬�ṹ������������������E�Ľṹ��ʽΪ
 ��
��A���ܷ���������Ӧ B���ұ����ϵ�һ�ȴ���ֻ��2��
��5�����Ȼ��۱���ϩ��
 ���Ǻϳɹ��ܸ߷��Ӳ��ϵ���Ҫ�м��壮
���Ǻϳɹ��ܸ߷��Ӳ��ϵ���Ҫ�м��壮��֪����NaBH4Ϊѡ���Ի�ԭ�������ܻ�ԭ����
�ڱ����ᷢ������ȡ��ʱ�����ɼ�λ���
��д���Լױ���Ϊ��Ҫԭ�ϣ��ϳɶ��Ȼ��۱���ϩ������ͼ�����Լ���ѡ�����ϳ�·������ͼʾ�����£�
CH3CH2OH$��_{170��}^{Ũ����}$CH2=CH2$\stackrel{Br_{2}}{��}$BrCH2-CH2Br��
���� �ݱ���ϩ�ĺϳ�·�߿�֪����������������ȡ����Ӧ����B����ͪ������ͪ������ԭ��Ӧ����C��C������ȥ��Ӧ���ɱ���ϩ���ݴ˷�����1����2����3����4����
��5���Լױ���Ϊ��Ҫԭ�ϣ��ϳɶ��Ȼ��۱���ϩʱ����Ҫ�ױ�������������ȡ����Ӧ���ɶԼ�����ͪ���������Ը�����������������Ȼ�����ԭͪ�����ǻ�������Ũ���������·�����ȥ��Ӧ����̼̼˫�������������Ӿ۷�Ӧ��ȡ ��
��
��� �⣺��1���ݱ���ϩ�ĺϳ�·�߿�֪����������������ȡ����Ӧ����B����ͪ��ͬʱ�������ᣬ�ʴ�Ϊ��ȡ����Ӧ��CH3COOH��
��2����C�Ľṹ��ʽ��֪��C�к����ǻ����ʴ�Ϊ���ǻ���
��3��C�к����ǻ���D��û�У�C�ܹ���Na��Ӧ�������壬���Լ������D������C��ʵ�鷽����ȡ���������������������壬˵������C��
�ʴ�Ϊ��ȡ���������������������壬˵������C��
��4����B�Ľṹ��ʽ��֪������ӽṹ�г��������2��̼ԭ�Ӻ�1����ԭ�ӣ��Ҵ���˫������ͬ���칹��E�ܷ���������Ӧ��˵������ȩ���������ϵ�һ�ȴ���ֻ��2�֣�˵���ڶ�λ�Ϸֱ�������ֲ�ͬ��ȡ�������ֱ�Ϊȩ���ͼ����ṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��5���Լױ���Ϊ��Ҫԭ�ϣ��ϳɶ��Ȼ��۱���ϩʱ����Ϊ�����ᷢ������ȡ��ʱ�����ɼ�λ�������������Ҫ�ױ�������������ȡ����Ӧ���ɶԼ�����ͪ��NaBH4Ϊѡ���Ի�ԭ�������ܻ�ԭ���ᣬ�������Ը�����������������Ȼ�����ԭͪ�����ǻ�������Ũ���������·�����ȥ��Ӧ����̼̼˫�������������Ӿ۷�Ӧ��ȡ ���ϳ�·������Ϊ
���ϳ�·������Ϊ $��_{��ˮ�Ȼ���}^{������}$
$��_{��ˮ�Ȼ���}^{������}$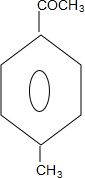 $\stackrel{���Ը������}{��}$
$\stackrel{���Ը������}{��}$ $\stackrel{NaBH_{4}}{��}$
$\stackrel{NaBH_{4}}{��}$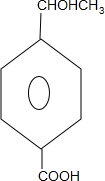 $��_{��}^{Ũ����}$
$��_{��}^{Ũ����}$ $\stackrel{������}{��}$
$\stackrel{������}{��}$ ��
��
�ʴ�Ϊ�� $��_{��ˮ�Ȼ���}^{������}$
$��_{��ˮ�Ȼ���}^{������}$ $\stackrel{���Ը������}{��}$
$\stackrel{���Ը������}{��}$ $\stackrel{NaBH_{4}}{��}$
$\stackrel{NaBH_{4}}{��}$ $��_{��}^{Ũ����}$
$��_{��}^{Ũ����}$ $\stackrel{������}{��}$
$\stackrel{������}{��}$ ��
��
���� ���⿼�����л���Ӧ�����Լ����л���Ĺ����Ž��������жϣ��ѵ����л��ϳ�·�ߵ���ƣ���Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д�| R | ||
| X | Y | Z |
| A�� | ��̬�⻯���ȶ��ԣ�Y��Z | |
| B�� | ԭ�Ӱ뾶��С˳����X��Y��R | |
| C�� | Y��R�γɵĻ�̨��YR2��ʹ����KMnO4��Һ��ɫ | |
| D�� | ����������ˮ���������X��Y��Z |
| A�� | CH3CH2Br$\stackrel{NaOHˮ��Һ}{��}$CH3CH2OH$\stackrel{Ũ���ᣬ170��}{��}$CH2=CH2$\stackrel{Br_{2}}{��}$CH2BrCH2Br | |
| B�� | CH3CH2Br$\stackrel{Br_{2}}{��}$CH2BrCH2Br | |
| C�� | CH3CH2Br$\stackrel{NaOH����Һ}{��}$CH2=CH2$\stackrel{Br_{2}}{��}$CH3CH2Br$\stackrel{Br_{2}}{��}$CH2BrCH2Br | |
| D�� | CH3CH2Br$\stackrel{NaOH����Һ}{��}$CH2=CH2$\stackrel{Br_{2}}{��}$CH2BrCH2Br |
| A�� | n��Cl2����n��Fe��=5��4������ 5Cl2+4Fe$\frac{\underline{\;��ȼ\;}}{\;}$2FeCl2+2FeCl3 | |
| B�� | n��Cl2����n��FeBr2��=1��1���� Fe2++2Br-+Cl2�TFe3++Br2+2Cl- | |
| C�� | n��MnO4-����n��H2O2��=2��3���� 2MnO4-+3H2O2+6H+�T2Mn2++4O2��+6H2O | |
| D�� | n��Fe����n��HNO3��ϡ�����T1��3�� 4Fe+12H++3NO3-=3Fe2++Fe3++3NO��+6H2O |
| A�� | �����������Ŀ�仯 | B�� | ��������Ӽ��仯 | ||
| C�� | һ������ | D�� | һ����С |
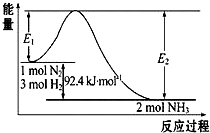 �ϳɰ���ҵ�ĺ��ķ�Ӧ�ǣ�N2��g��+3H2��g��?2NH3��g����H=��Q��kJ•mol-1�������仯��ͼ��ʾ���ش��������⣺
�ϳɰ���ҵ�ĺ��ķ�Ӧ�ǣ�N2��g��+3H2��g��?2NH3��g����H=��Q��kJ•mol-1�������仯��ͼ��ʾ���ش��������⣺