��Ŀ����
��ҵ�ϳ�����Ȼ����Ϊ�Ʊ�CH3OH��ԭ�ϡ���֪��
�� CH4(g)+O2(g)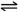 CO(g)+H2(g)+H2O(g) ��H=��321.5 kJ/mol
CO(g)+H2(g)+H2O(g) ��H=��321.5 kJ/mol
�� CH4(g)+ H2O(g) 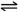 CO(g)+3H2(g) ��H=��250.
CO(g)+3H2(g) ��H=��250. 3 kJ/mol
3 kJ/mol
�� CO(g)+2H2(g) 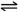 CH3OH(g) ��H=��90.0 kJ/mol
CH3OH(g) ��H=��90.0 kJ/mol
��1��CH4(g)��O2(g)��Ӧ����CH3OH(g)���Ȼ�ѧ����ʽΪ_________________________��
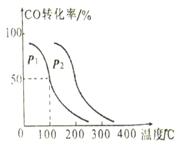
��2����V L�����ܱ������г���a mol CO��2a mol H2���ڲ�ͬѹǿ��
�ϳɼ״���CO ��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ʾ��
�� ѹǿP1____P2���������������������
�� ��100�桢P1ѹǿʱ��ƽ�ⳣ��Ϊ________ (�ú�a��V�Ĵ���ʽ��ʾ)��
��ϰ��ϵ�д�
�����Ŀ
���л����ij��ӷ����뷢����Ӧ�����;���ȷ����(����)
| ����� | �����Լ��� | ��Ӧ���� | |
| A | ��ȥO2�е�CO | ͨ����ȵ�����ͭ�� | ���Ϸ�Ӧ |
| B | ��ȥCO2�е�HCl | ͨ�뱥��̼��������Һ�� | ���ֽⷴӦ |
| C | ��ȥ̼����е�̼���� | �ܽ⡢�����Ȼ�����Һ | ���ֽⷴӦ |
| D | ��ȥ�Ȼ����е�̼���� | �����������ᡢ���� | ���ֽⷴӦ |


 Z��g��+2 W��g�� ��H<0�� ��Ӧ���е�5sʱ���X��ת����Ϊ25%��10 s��ﵽ��ѧƽ�⣬���Z��Ũ��Ϊ0.5mol/L��������˵����ȷ����
Z��g��+2 W��g�� ��H<0�� ��Ӧ���е�5sʱ���X��ת����Ϊ25%��10 s��ﵽ��ѧƽ�⣬���Z��Ũ��Ϊ0.5mol/L��������˵����ȷ����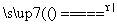 K2MnO4��MnO2��O2��
K2MnO4��MnO2��O2��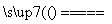 2NaCl
2NaCl