��Ŀ����
I���ڻ�ѧʵ��ʱ����ע�ⰲȫ���������������˺�������ʵ��������¹ʴ�������ȷ����______���ڵ�ȼ��ȼ����ǰ�������������Ĵ���
����ϡ��Ũ����ʱ��Ӧ��Ũ��������������ע��ˮ�У����ò���������
��Ũ�����Ƥ���и�ʴ�ԣ��粻��մ��Ƥ���ϣ�Ӧ�ý϶��ˮ��ϴ����Ϳ������ϡ��Һ �ܸ��Թ��е�Һ�����ʱ��Һ�岻�����Թ��ݻ���1/3
�ݵ�ȼ�����ƾ��ľƾ���
��ʵ��ʱ����������������ָ����������Ѫʱ�������Ȼ�����ҺͿĨֹѪ��ѪҺ�ɿ����ǽ����ɢϵ��II��ijͬѧ�õ�����������0.40mol/L��CuSO4��Һ240mL���ش���������
��1����������Ϊ��������ƽ��ҩ�ס��ձ�����ͷ�ιܣ�����Ҫ��Щ��������������ɸ�ʵ�飬��д����______��
��2����д����ʵ��ļ�Ҫ��ʵ�鲽�裺
�ټ��� �ڳ�������______g ��______��ת�� ��ϴ�Ӳ�ת�� ���� ��ҡ��
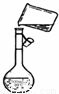
��3����ͼ�Ǹ�ͬѧת����Һ��ʾ��ͼ��ͼ�еĴ�����______��
��4����ͬѧ�ڶ��ݺӸǵ�תҡ�Ⱥ���Һ����ڿ̶��ߣ��ֵμ�����ˮ���̶ȣ���������ҺŨ�ȵ�Ӱ��______ ���ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��5��һƿ���ƺõ�ϡCuSO4��Һ����ʱ���������ǩ����Ϊ����������ʢ��CuSO4��Һ��˵˵���ʵ����鷽����
| ������� | ʵ����������� |
| Cu2+ | ______ |
| SO42- | ______ |
���𰸡�������I���ٿ�ȼ�������л���������ȼʱ�ᷢ����ը��
������Ũ������ܶȱ�ˮ����������ˮ������ˮʱ�ų��������ȣ�����ϡ��ʱһ��Ҫ��Ũ������������ˮ�У����ò��������裻
��Ũ�����Ƥ���и�ʴ�ԣ��粻��մ��Ƥ���ϣ�Ӧ�ý϶��ˮ��ϴ����Ϳ������ϡ��Һ��
�ܸ��ݸ��Թ��е�Һ����ȵķ������з����жϣ�
��װ���ƾ��ľƾ���������������ȼ�գ�
�����ڽ������Ӵ��е�ɣ��������ʻ��к͵�ɣ�ʹ��������۳���
II�� ��1������������Һ��ʵ���������ѡ�������������
��2��������n=cv���������ͭ�����ʵ���������ͭ�����غ��֪����ͭ������ͭ��������ʵ�����ȣ��ٸ���m=nM������������ͭ�����������
�۲��������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����
��3��ת����Һʱ����ֱ���㵹��Ӧ�ò�������������
��4���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=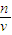 �����жϣ�
�����жϣ�
��5�����ݼ���Cu2+��SO42-�ķ�����
����⣺I���ٿ�ȼ�������л���������ȼʱ�ᷢ����ը���ʵ�ȼǰ������鴿�ȣ��ʢ���ȷ��
������Ũ������ܶȱ�ˮ����������ˮ������ˮʱ�ų��������ȣ�����ϡ��ʱһ��Ҫ��Ũ������������ˮ�У����ò��������裬�ʢ���ȷ��
��Ũ�����Ƥ���и�ʴ�ԣ��粻��մ��Ƥ���ϣ�Ӧ�ý϶��ˮ��ϴ����Ϳ������ϡ��Һ���ʢ۴���
�ܸ��Թ��е�Һ�����ʱ��Һ�岻�����Թ��ݻ���1/3���Է�ֹҺ�彦�����ʢ���ȷ��
��װ���ƾ��ľƾ���������������ȼ�գ��ʢݴ���
�����ڽ������Ӵ��е�ɣ��������ʻ��к͵�ɣ�ʹ��������۳������������Ȼ�����ҺͿĨֹѪ���ʢ���ȷ��
��ѡ���ۢݣ�
II�� ��1�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣮��ȴ��ת�Ƶ�250mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ�����������������ƽ���ձ�����������250mL����ƿ����ͷ�ιܡ�ҩ�ף��ʴ𰸣�250mL����ƿ����������
��2����������ͭ���������Ϊm=0.25L×0.4mol?L-1×250g/mol=2.5g���ʴ�Ϊ��2.5��
�۲��������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ������ʴ�Ϊ���ܽ⣻
��3��ת����Һʱ����ֱ���㵹��Ӧ�ò��������������ʴ�Ϊ��ת����Һʱ����ֱ���㵹��Ӧ�ò�������������
��4��ҡ�Ⱥ�Һ���½���һ������Һ����ƿ����ƿ��֮�䣬�ټ�����ˮ���̶��ߣ�������Һ���ƫ��������ҺŨ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��5������Cu2+�ķ�����ȡ����������Һ���Թ��У����뼸��NaOH��Һ������ɫ�������ɣ�����SO42-�ķ�����ȡ����������Һ���Թ��У����������ữ���ٵ��뼸��BaCl2��Һ���а�ɫ�������ɣ��ʴ�Ϊ��ȡ����������Һ���Թ��У����뼸��NaOH��Һ������ɫ�������ɣ�ȡ����������Һ���Թ��У����������ữ���ٵ��뼸��BaCl2��Һ���а�ɫ�������ɣ�
���������⿼���˳�����ʵ������Լ�һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ���ע��û��240mL������ƿ����Һʵ��Ӧ����250ml�����ù��������Ӧ������250mL��Һ�������õ���������
������Ũ������ܶȱ�ˮ����������ˮ������ˮʱ�ų��������ȣ�����ϡ��ʱһ��Ҫ��Ũ������������ˮ�У����ò��������裻
��Ũ�����Ƥ���и�ʴ�ԣ��粻��մ��Ƥ���ϣ�Ӧ�ý϶��ˮ��ϴ����Ϳ������ϡ��Һ��
�ܸ��ݸ��Թ��е�Һ����ȵķ������з����жϣ�
��װ���ƾ��ľƾ���������������ȼ�գ�
�����ڽ������Ӵ��е�ɣ��������ʻ��к͵�ɣ�ʹ��������۳���
II�� ��1������������Һ��ʵ���������ѡ�������������
��2��������n=cv���������ͭ�����ʵ���������ͭ�����غ��֪����ͭ������ͭ��������ʵ�����ȣ��ٸ���m=nM������������ͭ�����������
�۲��������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����
��3��ת����Һʱ����ֱ���㵹��Ӧ�ò�������������
��4���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=
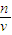 �����жϣ�
�����жϣ���5�����ݼ���Cu2+��SO42-�ķ�����
����⣺I���ٿ�ȼ�������л���������ȼʱ�ᷢ����ը���ʵ�ȼǰ������鴿�ȣ��ʢ���ȷ��
������Ũ������ܶȱ�ˮ����������ˮ������ˮʱ�ų��������ȣ�����ϡ��ʱһ��Ҫ��Ũ������������ˮ�У����ò��������裬�ʢ���ȷ��
��Ũ�����Ƥ���и�ʴ�ԣ��粻��մ��Ƥ���ϣ�Ӧ�ý϶��ˮ��ϴ����Ϳ������ϡ��Һ���ʢ۴���
�ܸ��Թ��е�Һ�����ʱ��Һ�岻�����Թ��ݻ���1/3���Է�ֹҺ�彦�����ʢ���ȷ��
��װ���ƾ��ľƾ���������������ȼ�գ��ʢݴ���
�����ڽ������Ӵ��е�ɣ��������ʻ��к͵�ɣ�ʹ��������۳������������Ȼ�����ҺͿĨֹѪ���ʢ���ȷ��
��ѡ���ۢݣ�
II�� ��1�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ�����ձ��������ò��������裬�����ܽ⣮��ȴ��ת�Ƶ�250mL����ƿ�У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ�����������������ƽ���ձ�����������250mL����ƿ����ͷ�ιܡ�ҩ�ף��ʴ𰸣�250mL����ƿ����������
��2����������ͭ���������Ϊm=0.25L×0.4mol?L-1×250g/mol=2.5g���ʴ�Ϊ��2.5��
�۲��������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ������ʴ�Ϊ���ܽ⣻
��3��ת����Һʱ����ֱ���㵹��Ӧ�ò��������������ʴ�Ϊ��ת����Һʱ����ֱ���㵹��Ӧ�ò�������������
��4��ҡ�Ⱥ�Һ���½���һ������Һ����ƿ����ƿ��֮�䣬�ټ�����ˮ���̶��ߣ�������Һ���ƫ��������ҺŨ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��5������Cu2+�ķ�����ȡ����������Һ���Թ��У����뼸��NaOH��Һ������ɫ�������ɣ�����SO42-�ķ�����ȡ����������Һ���Թ��У����������ữ���ٵ��뼸��BaCl2��Һ���а�ɫ�������ɣ��ʴ�Ϊ��ȡ����������Һ���Թ��У����뼸��NaOH��Һ������ɫ�������ɣ�ȡ����������Һ���Թ��У����������ữ���ٵ��뼸��BaCl2��Һ���а�ɫ�������ɣ�
���������⿼���˳�����ʵ������Լ�һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ���ע��û��240mL������ƿ����Һʵ��Ӧ����250ml�����ù��������Ӧ������250mL��Һ�������õ���������

��ϰ��ϵ�д�
�����Ŀ
 I���ڻ�ѧʵ��ʱ����ע�ⰲȫ���������������˺�������ʵ��������¹ʴ�������ȷ����
I���ڻ�ѧʵ��ʱ����ע�ⰲȫ���������������˺�������ʵ��������¹ʴ�������ȷ����



 I���ڻ�ѧʵ��ʱ����ע�ⰲȫ���������������˺�������ʵ��������¹ʴ�������ȷ����______
I���ڻ�ѧʵ��ʱ����ע�ⰲȫ���������������˺�������ʵ��������¹ʴ�������ȷ����______