��Ŀ����
(12��)��������Ϊԭ�������������õ����Է�ˮ����Ԫ�غ������ߣ�Ϊ��������ŷţ����û�ѧ���������������ˮ������������±���
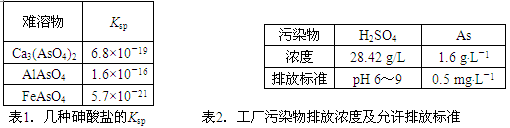
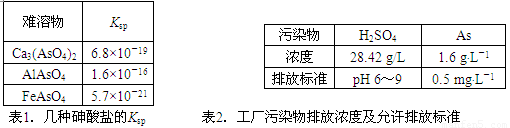
��1�����������ε�Ksp ��2��������Ⱦ���ŷ�Ũ�ȼ������ŷű�
�ش��������⣺
�Ÿ����Ṥ���ŷŵķ�ˮ����������ʵ���Ũ��c(H2SO4)��______________mol��L��1��
��д��������Ca3(AsO4)2��Ksp����ʽ��Ksp[Ca3(AsO4)2]��______________���������Һ
��Al3����Fe3����Ũ�Ⱦ�Ϊ1.0��10��4mol��L��1��c(AsO43��)�������_______________mol��L��1��
�ǹ����ŷų������Է�ˮ�е������飨H3AsO3���ᣩ���׳�������Ͷ��MnO2�Ƚ�������������飨H3AsO4���ᣩ��д���÷�Ӧ�����ӷ���ʽ_________________________��
���ڴ��������ˮʱ���÷ֶ�ʽ�������ˮ��Ͷ����ʯ�ҵ���pH��2����Ͷ����ʯ�ҽ�pH���ڵ�8����ʹ�������Ca3(AsO4)2��ʽ������
�ٽ�pH���ڵ�2ʱ��ˮ���д�������������������Ҫ�ɷֵĻ�ѧʽΪ______________��
��Ca3(AsO4)2��pH���ڵ�8���Ҳſ�ʼ������ԭ��Ϊ_____________________________��
��12�֣���0.29
��c3 (Ca2��)��c2(AsO43��) 5.7��10��17
��2H����MnO2��H3AsO3��H3AsO4��Mn2����H2O
�Ȣ�CaSO4
��H3AsO4�����ᣬ����Һ��pH���ڵ�8����ʱAsO43��Ũ������Ca3(AsO4)2��ʼ������ÿ��2�֣���12�֣�
����:

 ��У����ϵ�д�
��У����ϵ�д�(12��)��������Ϊԭ�������������õ����Է�ˮ����Ԫ�غ������ߣ�Ϊ��������ŷţ����û�ѧ���������������ˮ������������±���
| ������ | Ksp |
| Ca3(AsO4)2 | 6.8��10��19 |
| AlAsO4 | 1.6��10��16 |
| FeAsO4 | 5.7��10��21 |
��1�����������ε�Ksp
| ��Ⱦ�� | H2SO4 | As |
| Ũ�� | 28.42 g/L | 1.6 g��L��1 |
| �ŷű� | pH 6��9 | 0.5 mg��L��1 |
��2��������Ⱦ���ŷ�Ũ�ȼ������ŷű�
�ش��������⣺
�Ÿ����Ṥ���ŷŵķ�ˮ����������ʵ���Ũ��c(H2SO4)��______________mol��L��1��
��д��������Ca3(AsO4)2��Ksp����ʽ��Ksp[Ca3(AsO4)2]��______________���������Һ
��Al3����Fe3����Ũ�Ⱦ�Ϊ1.0��10��4mol��L��1��c(AsO43��)�������_______________mol��L��1��
�ǹ����ŷų������Է�ˮ�е������飨H3AsO3���ᣩ���׳�������Ͷ��MnO2�Ƚ�������
������飨H3AsO4���ᣩ��д���÷�Ӧ�����ӷ���ʽ________________________________��
���ڴ��������ˮʱ���÷ֶ�ʽ�������ˮ��Ͷ����ʯ�ҵ���pH��2����Ͷ����ʯ�ҽ�pH
���ڵ�8����ʹ�������Ca3(AsO4)2��ʽ������
�ٽ�pH���ڵ�2ʱ��ˮ���д�������������������Ҫ�ɷֵĻ�ѧʽΪ______________��
��Ca3(AsO4)2��pH���ڵ�8���Ҳſ�ʼ������ԭ��Ϊ_____________________________��
(12��)��������Ϊԭ�������������õ����Է�ˮ����Ԫ�غ������ߣ�Ϊ��������ŷţ����û�ѧ���������������ˮ������������±���
| ������ | Ksp |
| Ca3(AsO4)2 | 6.8��10��19 |
| AlAsO4 | 1.6��10��16 |
| FeAsO4 | 5.7��10��21 |
| ��Ⱦ�� | H2SO4 | As |
| Ũ�� | 28.42 g/L | 1.6 g��L��1 |
| �ŷű� | pH 6��9 | 0.5 mg��L��1 |
�ش��������⣺
�Ÿ����Ṥ���ŷŵķ�ˮ����������ʵ���Ũ��c(H2SO4)��______________mol��L��1��
��д��������Ca3(AsO4)2��Ksp����ʽ��Ksp[Ca3(AsO4)2]��______________���������Һ
��Al3����Fe3����Ũ�Ⱦ�Ϊ1.0��10��4mol��L��1��c(AsO43��)�������_______________mol��L��1��
�ǹ����ŷų������Է�ˮ�е������飨H3AsO3���ᣩ���׳�������Ͷ��MnO2�Ƚ�������������飨H3AsO4���ᣩ��д���÷�Ӧ�����ӷ���ʽ________________________________��
���ڴ��������ˮʱ���÷ֶ�ʽ�������ˮ��Ͷ����ʯ�ҵ���pH��2����Ͷ����ʯ�ҽ�pH
���ڵ�8����ʹ�������Ca3(AsO4)2��ʽ������
�ٽ�pH���ڵ�2ʱ��ˮ���д�������������������Ҫ�ɷֵĻ�ѧʽΪ______________��
��Ca3(AsO4)2��pH���ڵ�8���Ҳſ�ʼ������ԭ��Ϊ_____________________________��
 �ش��������⣺
�ش��������⣺