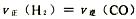��Ŀ����
�ö��Ե缫���һ��������ijŨ�ȵ�NaCl��Һ��һ��ʱ���ֹͣ��⡣��ʱ������100g36.5%��Ũ���ᣬ������Һ������ԭ��Һ��ȫ��ͬ��������˵����ȷ���ǣ�����
A���������У��������õ������壬����ͬ��������������
B���������У�����ͬ�������£��������õ������������������Ĵ�
C����������ת�Ƶ��ӵ����ʵ���ԼΪ8mol
D��ԭijŨ�ȵ�NaCl��Һ������117gNaCl
C
����:
���NaCl��Һ����貹��HCl���ɸ�ԭ���ּ���100g36.5%��Ũ���ᣬn��HCl��=1mol��n��H2O��=3.5mol����HCl��NaOH=NaCl��H2O֪��ԭNaCl��Һ��58.5gNaCl��D����ȷ��������������һ����2NaCl��2H2O![]() Cl2����H2����2NaOH��ת��1mol���ӣ���������Cl20.5mol����������H20.5mol���ڶ�����2H2O
Cl2����H2����2NaOH��ת��1mol���ӣ���������Cl20.5mol����������H20.5mol���ڶ�����2H2O![]() O2����2H2��ת�Ƶ���7mol����������O21.75mol����������H23.5mol�����Ϸ�������������Cl2��O2��2.25mol����������H2��4mol����A��B����ȷ���ܹ�ת�Ƶ��ӵ����ʵ���n��e����=1mol��7mol=8mol��
O2����2H2��ת�Ƶ���7mol����������O21.75mol����������H23.5mol�����Ϸ�������������Cl2��O2��2.25mol����������H2��4mol����A��B����ȷ���ܹ�ת�Ƶ��ӵ����ʵ���n��e����=1mol��7mol=8mol��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ