��Ŀ����
�ظ���أ�K2Cr2O7)�Ǹ��л�ѧ����������������ҵ���Ը�����Ϊԭ���ü����������Ʊ�����������ͨ������Cr2O3��FeO��Al2O3��SiO2�ȡ�
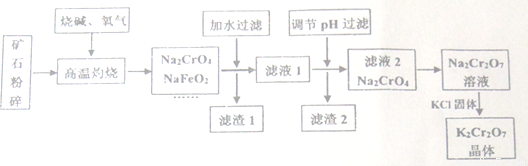
��֪����NaFeO2��ˮǿ��ˮ���2CrO42-(��ɫ) + 2H+ Cr2O72-(��ɫ)+H2O
Cr2O72-(��ɫ)+H2O
��ش��������⣺
(1)����ʯ�����Ŀ����________����������ʱCr2O3������Ӧ�Ļ�ѧ����ʽΪ__________��
(2)����1���к��ɫ���ʣ�д�����ɸ����ʷ�Ӧ�����ӷ���ʽ________������2����Ҫ�ɷ���A1(OH)3��______________��
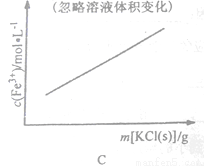
(3)�ü�Ҫ������˵��Na2Cr2O7��Һ�м���KC1���壬��������K2Cr2O7��ԭ��_______��
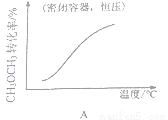
(4) 25��Cʱ���Է�Ӧ2CrO42-(��ɫ)+2H+ Cr2O72- (��ɫ)+H2O��ȡNa2CrO4��Һ����ʵ�飬��ò���ʵ���������£�
Cr2O72- (��ɫ)+H2O��ȡNa2CrO4��Һ����ʵ�飬��ò���ʵ���������£�
ʱ��/ (S) | 0 | 0.01 | 0.02 | 003 | 0.04 |
(CrO42-)/ (mol��L-1) | 0.20 | 1.6��10-2 | 1.2��10-2 | 1.0��10-2 | |
(Cr2O72-)/ (mol��L-1) | 0 | 9.2��l0-2 | 9.4��10-2 | 9.5��10-2 |
��Ӧ�ﵽƽ��ʱ����Һ��pH=l���÷�Ӧƽ�ⳣ��KΪ______��
�������й�˵����ȷ��_____________��
a.������NaHCO3���壬��ʹ��Һ�ij�ɫ����
b.0.03 sʱv(CrO42-)(��)=2v(Cr2O72-)(��)
c.��Һ��c(CrO42-):c(Cr2O72-)=2 : 1ʱ�÷�Ӧ����ƽ��״̬
d.��Ӧ�ﵽƽ��ʱCrO42-��ת����Ϊ95%
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�

 ��˵����ȷ���ǣ� ��
��˵����ȷ���ǣ� �� ����������������֮��Ϊ50 D.
����������������֮��Ϊ50 D. 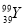 ��
��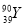 �����ֲ�ͬ�ĺ���
�����ֲ�ͬ�ĺ���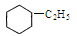 ����һ����ﹲ�м��֣������������칹��
����һ����ﹲ�м��֣������������칹�� 6H2(g) + 2CO2(g)�� ��H>0
6H2(g) + 2CO2(g)�� ��H>0