��Ŀ����
��1����0.2mol/L HA ��Һ�� 0.1mol/L NaOH��Һ�������ϣ���û����Һ��c��Na+��>c��A?�������á�>������<����=����д���пհף���
�ٻ����Һ��c��A?��_________c��HA����
�ڻ����Һ��c��HA�� + c��A?�� 0.1mol/L��
��2������ʱ��ȡ0.1mol•L-1 HX��Һ��0.1mol•L-1 NaOH��Һ�������ϣ���Ϻ���Һ����ı仯���ƣ���û����Һ��pH=8���Իش��������⣺
�ٻ����Һ����ˮ�������c��OH-����0.1mol•L-1 NaOH��Һ����ˮ�������c��OH-��֮��Ϊ ��
����֪NH4X��Һ�����ԣ���֪��HX��Һ���뵽Na2CO3��Һ��������ų������ƶϣ�NH4��2CO3��Һ��pH__________7��ѡ���������������������
��ϰ��ϵ�д�
�����Ŀ


 2C��g���ﵽƽ��ʱ��A��B��C�����ʵ����ֱ�Ϊ4moL��2moL��4moL�������¶Ⱥ�ѹǿ���䣬��ƽ�����������ߵ��������µ�������ʹƽ�����Ƶ��ǣ� ��
2C��g���ﵽƽ��ʱ��A��B��C�����ʵ����ֱ�Ϊ4moL��2moL��4moL�������¶Ⱥ�ѹǿ���䣬��ƽ�����������ߵ��������µ�������ʹƽ�����Ƶ��ǣ� �� �� C��������1moL D��������1moL
�� C��������1moL D��������1moL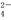 �����ʵ���Ũ���� ( )
�����ʵ���Ũ���� ( ) C������
C������