��Ŀ����
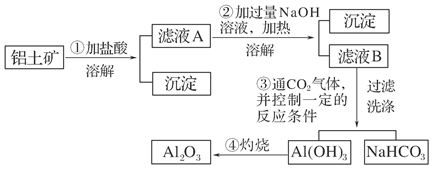
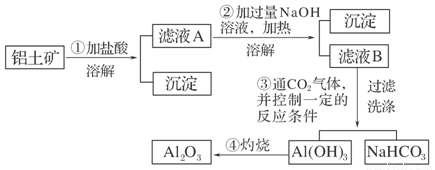
(11��)ij̽��С����ʵ��������������(��Ҫ�ɷ�ΪAl2O3��������Fe2O3��SiO2)��ȡ���������ش��������⣺
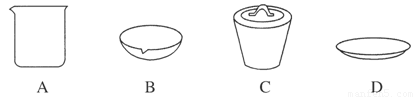
(1)��ʵ��������1 mol��L��1��NaOH��Һ480 mL�����Ƹ���Һ��������������������ƽ(����)����ͷ�ιܡ�ҩ�ס�����������ȱ�ٵ�������_______________________��
�����ղ������õ����������е�һ�֣���������________��
(2)д��������з�����Ӧ�����ӷ���ʽ____________________________________
________________________________________________________________________��
(3)��������ϴ����β���_______________________________________________
________________________________________________________________________��
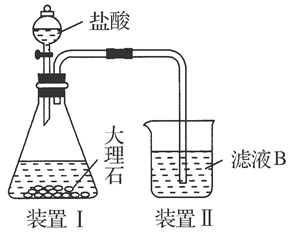
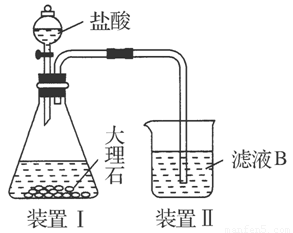
(4)��ͬѧ��ʵ������������װ���Ʊ�CO2���壬��ͨ����ҺB����ȡAl(OH)3ʱ�����û�в���Ԥ������
��ͬѧ������Ϊ����ͬѧͨ��CO2�����ǵ���ʵ��ʧ�ܵ�ԭ��֮һ������Ϊ�ҵķ����Ƿ������________�����������������ӷ���ʽ������ԭ��________________________________________________________��
(������Ϊ���������ÿղ�����)
��ͬѧ������Ϊ����ͬѧͨ���CO2�к���HCl���壬Ҳ�ǵ���ʵ��ʧ�ܵ�ԭ����ʵ��������ijװ�ÿɽ��������⡣�������ͬѧ������װ��ͼ����ע���Լ����ơ�

(1)500 mL����ƿ���ձ�'����
(2)Fe2O3��6H��===2Fe3����3H2O��Al2O3��6H��===2Al3����3H2O
(3)��������м�������ˮ��ʹˮ�պ�û���������ˮ�������ظ�����2��3��
(4)����'2OH����CO2===CO32����H2O
��װ�â��װ�â�֮���һϴ��ƿ��װ�����¡�
����:��������������ܽ����ΪSiO2����ҺA��AlCl3��FeCl3�����Һ���ټ������NaOH��Һ���Fe(OH)3��������ҺB��NaAlO2��ҺB��ͨ�����CO2�Ϳ�����Al(OH)3������NaHCO3��

 ��У����ϵ�д�
��У����ϵ�д���1��(4��)ij̽��С����HNO3�����ʯ��Ӧ������������С�ķ������о�Ӱ�췴Ӧ���ʵ����ء�����HNO3Ũ��Ϊ1.00mol��L��2.00mol��L������ʯ��ϸ������ֿ������ֹ��ʵ���¶�Ϊ298K��308K��ÿ��ʵ��HNO3������Ϊ25.0mL������ʯ����Ϊ10.00g�����������ʵ����Ʊ��������ʵ��Ŀ��һ���е���գ�
| ʵ�� ��� | T/K | ����ʯ ��� | HNO3Ũ�� /(mol/L) | ʵ �� Ŀ �� |
| �� | 298 | �ֿ��� | 2.00 | (I)ʵ��ٺ͢�̽��HNO3Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죻 (��)ʵ��ٺ͢�̽��________________�Ը÷�Ӧ���ʵ�Ӱ�죻 (��)ʵ��ٺ͢�̽��________________�Ը÷�Ӧ���ʵ�Ӱ�졣 |
| �� | 298 | �ֿ��� | 1.00 | |
| �� | 298 | ϸ���� | 2.00 | |
| �� | 308K | �ֿ��� | 2.00 |
��2����4�֣���11.2L����״������ϩ������Ļ������ͨ��������������Ȼ�̼��Һ�У���ַ�Ӧ��������Ȼ�̼��Һ������������8.4g����ԭ������������ϩ����������ʵ���֮�Ⱥ������ȡ�
��1��(4��)ij̽��С����HNO3�����ʯ��Ӧ������������С�ķ������о�Ӱ�췴Ӧ���ʵ����ء�����HNO3Ũ��Ϊ1.00mol��L��2.00mol��L������ʯ��ϸ������ֿ������ֹ��ʵ���¶�Ϊ298K��308K��ÿ��ʵ��HNO3������Ϊ25.0mL������ʯ����Ϊ10.00g�����������ʵ����Ʊ��������ʵ��Ŀ��һ���е���գ�
|
ʵ�� ��� |
T/K |
����ʯ ��� |
HNO3Ũ�� /(mol/L) |
ʵ �� Ŀ �� |
|
�� |
298 |
�ֿ��� |
2.00 |
(I)ʵ��ٺ͢�̽��HNO3Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죻 (��)ʵ��ٺ͢�̽��________________�Ը÷�Ӧ���ʵ�Ӱ�죻 (��)ʵ��ٺ͢�̽��________________�Ը÷�Ӧ���ʵ�Ӱ�졣 |
|
�� |
298 |
�ֿ��� |
1.00 |
|
|
�� |
298 |
ϸ���� |
2.00 |
|
|
�� |
308K |
�ֿ��� |
2.00 |
��2����4�֣���11.2L����״������ϩ������Ļ������ͨ��������������Ȼ�̼��Һ�У���ַ�Ӧ��������Ȼ�̼��Һ������������8.4g����ԭ������������ϩ����������ʵ���֮�Ⱥ������ȡ�