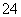��Ŀ����
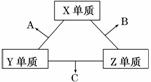
������Ԫ����Ԫ����ռ����Ҫ��λ��X��Y��Z���ֶ�����Ԫ�أ����ǵ�ԭ������֮��Ϊ16��X��Y��Z����Ԫ�صij��������ڳ����¶�����ɫ���壬���ʵ������¿ɷ����� �±仯��
�±仯��
 ��
��
һ��B�����к��е�Zԭ�Ӹ�����C��������1����B��C���ַ����еĵ�����������10����ش��������⣺
(1)XԪ�������ڱ��е�λ����____________����________�塣
(2)����ͬ����Ԫ�����ʵĵݱ���ɣ�����B��C���ʷе㷴����������Ϊ���ǵķ���֮�����________��
(3)��C�����У�Yԭ�ӵ��ӻ���ʽΪ________����Yԭ��ͬ�����ڵ������ڵ�Ԫ��ԭ�ӻ�̬�����Ų�ʽ��____________________________________________________________��
(4)NaCl�ľ���ṹ��ͼ��ʾ�������������ĺ����ĵ�ԭ�ӳ�ȥ������������ӻ�ΪXn��(X����ijԪ�ط���)���ӣ����ϵ�Cl����ΪO2�����͵õ�X��������ľ���ṹ�����X��������Ļ�ѧʽΪ___________________________________________________��
������(1)�����ڳ��������嵥��X��Z��Ӧ����10���ӻ�����B��Y������Z���ʷ�Ӧ����10���ӻ�����C����һ��B�����к��е�Zԭ�Ӹ�����C��������1������CΪNH3��BΪH2O��XΪ��Ԫ�أ�YΪ��Ԫ�أ�ZΪ��Ԫ�أ���Ԫ��λ��Ԫ�����ڱ��ڶ����ڵڢ�A�塣
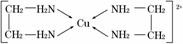
(2)ˮ���Ӽ䡢�����Ӽ����γ�������� ��H2O��NH3�ķе���ַ�����
��H2O��NH3�ķе���ַ�����
(3)�������ڵڢ�A���Ԫ����As�����̬ԭ�ӵĵ����Ų�Ϊ1s22s22p63s23p63d104s24p3��
(4)�����к�Xn����O2������Ŀ ֮��Ϊ��8��
֮��Ϊ��8�� ��12��
��12�� ��1��3����X��������Ļ�ѧʽΪXO3��
��1��3����X��������Ļ�ѧʽΪXO3��
�𰸣�(1)������A��(2)���
(3)sp3��1s22s22p63s23p63d104s24p3��(4)XO3

 �Ǽ�����������ϵ�д�
�Ǽ�����������ϵ�д� â���̸������Ծ�ϵ�д�
â���̸������Ծ�ϵ�д�Ϊ�˽����±��Т����е�ʵ�飬��Ӣ�����ѡ��һ���Լ����Ӣ�ѡ��һ�ֺ��ʵ�ʵ������������Ѵ�д���±��еĴ����ڣ�
| ��ʵ�� | ���Լ� | ��������� | �� | ||
| (1)��ȥ���еı��� | A.������ | a.��Һ | (1) | ||
| (2)��ȥ��ҵ�ƾ��е�ˮ | B.����̼�� ����Һ | b.���� | (2) | ||
| (3)��ȥ���������е����� | C.�Ȼ��� | c.���� | (3) | ||
| (4)��ȥ�����еĸ��� | D.�������� | d.���� | (4) |
 2CuCl����4H����SO
2CuCl����4H����SO