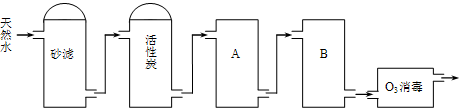
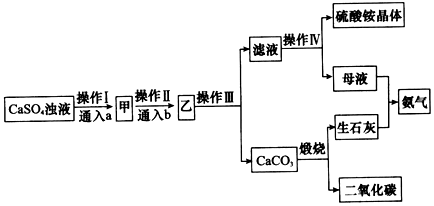
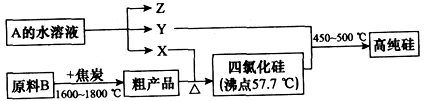
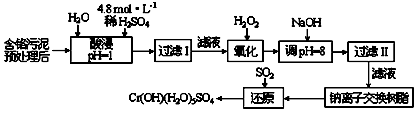
��Ŀ����
����ѡһ������ѧ�뼼����
���Ṥҵ����Ӧ�����ۺϾ���Ч�����⡣
(1)���������ĸ�������ѡ��һ���½�һ�����᳧������Ϊ��ַ��ѡ��_________�����ţ��Ľ�����
A���зḻ��������Դ�ij���
B����������������
C��������������Ĺ�ҵ����
D���˿ڳ��ܵ��Ļ�����ҵ���ij���
(2)�ݲ��㣬�Ӵ�������������У�����Ӧ�ȶ�δ�����ã���ÿ����1t98%������������3.6��10kJ��������ͨ�������жϣ�����Ӧ��SO2(g)+O2(g) SO3(g) ��H=-98.3kJ/mol�ų��������������������еõ�������ã���ÿ����1t98%������ֻ������ṩ���������������____kJ������
SO3(g) ��H=-98.3kJ/mol�ų��������������������еõ�������ã���ÿ����1t98%������ֻ������ṩ���������������____kJ������
(3)CuFeS2�ǻ��������һ�ɷ֣�����ʱ��CuFeS2ת��ΪCuO��Fe2O3��SO2���÷�Ӧ�Ļ�ѧ����ʽΪ___________________��
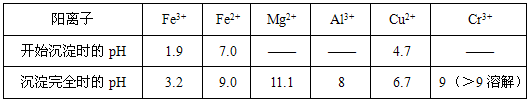
(4)�����᳧����¯�ų��Ŀ����к���Fe2O3��CuO��CuSO4 ����CuO��SO3�ڷ���¯�л��϶��ɣ�����������ͭ���������������¯�¶Ȳ�ͬ���仯�����±�����
���Ṥҵ����Ӧ�����ۺϾ���Ч�����⡣
(1)���������ĸ�������ѡ��һ���½�һ�����᳧������Ϊ��ַ��ѡ��_________�����ţ��Ľ�����
A���зḻ��������Դ�ij���
B����������������
C��������������Ĺ�ҵ����
D���˿ڳ��ܵ��Ļ�����ҵ���ij���
(2)�ݲ��㣬�Ӵ�������������У�����Ӧ�ȶ�δ�����ã���ÿ����1t98%������������3.6��10kJ��������ͨ�������жϣ�����Ӧ��SO2(g)+O2(g)
 SO3(g) ��H=-98.3kJ/mol�ų��������������������еõ�������ã���ÿ����1t98%������ֻ������ṩ���������������____kJ������
SO3(g) ��H=-98.3kJ/mol�ų��������������������еõ�������ã���ÿ����1t98%������ֻ������ṩ���������������____kJ������ (3)CuFeS2�ǻ��������һ�ɷ֣�����ʱ��CuFeS2ת��ΪCuO��Fe2O3��SO2���÷�Ӧ�Ļ�ѧ����ʽΪ___________________��
(4)�����᳧����¯�ų��Ŀ����к���Fe2O3��CuO��CuSO4 ����CuO��SO3�ڷ���¯�л��϶��ɣ�����������ͭ���������������¯�¶Ȳ�ͬ���仯�����±�����

��֪CuSO4�ڵ���660��ʱ����ֽ⣬���Ҫ�����ϱ���CuSO4�������������¶����߶����͵�ԭ����_____________________
(1)C
(2)6.23��105
(3)4CuFeS2+13O2 4CuO+2Fe2O3+8SO2
4CuO+2Fe2O3+8SO2
(4)SO2ת��ΪSO3������Ӧ���ȵĿ��淴Ӧ�����¶����ߣ�ƽ�������ƶ���SO3�����ʵ������٣�����CuSO4�������٣����¶����ߣ�SO3�����ʵ������٣���CuSO4�������٣�
(2)6.23��105
(3)4CuFeS2+13O2
 4CuO+2Fe2O3+8SO2
4CuO+2Fe2O3+8SO2 (4)SO2ת��ΪSO3������Ӧ���ȵĿ��淴Ӧ�����¶����ߣ�ƽ�������ƶ���SO3�����ʵ������٣�����CuSO4�������٣����¶����ߣ�SO3�����ʵ������٣���CuSO4�������٣�

��ϰ��ϵ�д�
 ���ʿ��ÿ��ֳɳ�ϵ�д�
���ʿ��ÿ��ֳɳ�ϵ�д�
�����Ŀ
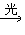 CH3CH2Cl��HCl
CH3CH2Cl��HCl 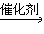 CH3CH2Cl
CH3CH2Cl