��Ŀ����
��1������ʵ���еĻ�ѧ����ʽ��
��2������ͭ��Һ���Լӿ������������ʵ�ԭ����
��3��ʵ����������Na2SO4��Ag2SO4��MgSO4��K2SO4��4����Һ������ʵ����CuSO4��Һ���������õ���
��4��Ҫ�ӿ�����ʵ����������������ʣ����ɲ�ȡ�Ĵ�ʩ�У���һ�ּ��ɣ���
��2��Zn������ķ�Ӧ�м���������CuSO4��Һ�����û���һ����Cu������Һ���γ�Cu/Znԭ��أ�ԭ��ط�Ӧ�Ȼ�ѧ��Ӧ���ʿ죻
��3������������Һ��ֻZnֻ���û���Ag��
��4����Ӱ�췴Ӧ���ʵ����ط�����
�ʴ�Ϊ��CuSO4+Zn=ZnSO4+Cu��Zn+H2SO4=ZnSO4+H2������
��2��Zn������ķ�Ӧ�м���������CuSO4��Һ�����û���һ����Cu������Һ���γ�Cu/Znԭ��أ����ӿ컯ѧ��Ӧ���ʣ�
�ʴ�Ϊ��CuSO4��Zn��Ӧ������Cu��Zn�γ�Cu-Znԭ��أ��ӿ����������ɵ����ʣ�
��3������������Һ��ֻZnֻ���û���Ag����Ag2SO4��CuSO4��Һ�������Ƶ����ã��ʴ�Ϊ��Ag2SO4��
��4��Ϊ�ӿ�Zn������ķ�Ӧ���ʿɴ����¡��ʵ����������Ũ�ȡ�����п�ıȱ�����ȽǶȿ��ǣ�
�ʴ�Ϊ������п�ı�����������¶Ȼ��ʵ����������Ũ�ȵȣ���һ�ּ��ɣ���

ijͬѧ����ϡ������п��ȡ������ʵ���У����ּ�����������ͭ��Һ�ɼӿ��������������ʡ���ش��������⣺
(1)����ʵ���з�����Ӧ�Ļ�ѧ����ʽ��_________________________________________��
__________________________________________��
(2)��������ͭ��Һ����Լӿ������������ʵ�ԭ����_____________________________��
(3)ʵ����������Na2SO4��MgSO4��Ag2SO4��K2SO4��4����Һ����������ʵ����CuSO4��Һ���������õ���________��
(4)Ҫ�ӿ�����ʵ���������������ʣ����ɲ�ȡ�Ĵ�ʩ��___________________________
____________________________________________________________ (�����ּ���)��
(5)Ϊ�˽�һ���о�����ͭ�����������������ʵ�Ӱ�죬��ͬѧ���������һϵ�е�ʵ�飺
|
���� ʵ�� �����Һ������ |
A |
B |
C |
D |
E |
F |
|
4 mol/L H2SO4��mL�� |
30 |
V1 |
V2 |
V3 |
V4 |
V5 |
|
����CuSO4��Һ��mL�� |
0 |
0.5 |
2.5 |
5 |
V6 |
20 |
|
H2O��mL�� |
V7 |
V8 |
V9 |
V10 |
10 |
0 |
�����������Ļ����Һ�ֱ���뵽6��ʢ�й���Zn���������У��ռ����������壬��¼�����ͬ�������������ʱ�䡣
������ɴ�ʵ����ƣ����У�V1��__________��V6��__________��V9��________��
�ڷ�Ӧһ��ʱ���ʵ��A�еĽ�����________ɫ��ʵ��E�еĽ�����________ɫ��
�۸�ͬѧ���ó��Ľ���Ϊ������������CuSO4��Һʱ���������������ʻ�����ߣ����������CuSO4��Һ����һ����ʱ���������������ʷ������½���������������������½�����Ҫԭ��_____________________________________________________________________________________________________________________________��
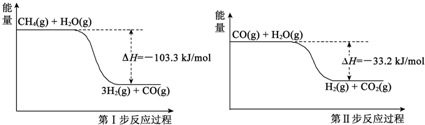 ������һ�������Դ����������ȡ�봢��������Դ����������о��ȵ㣮
������һ�������Դ����������ȡ�봢��������Դ����������о��ȵ㣮 ��
�� ��
�� ��
�� ��4����Һ������ʵ����
��4����Һ������ʵ���� ��Һ���������õ���
��
��Һ���������õ���
��