��Ŀ����
��10�֣���1��ij��ѧ�����о�С�飬���ʵ��̽��KI��Һ��FeCl3��Һ��Ӧ����һ�����ȡ���֪�÷�ӦΪ��2Fe3+ +2I- ��2Fe2+ + I2���������ص�ʵ�鲽�������
��ѡ�Լ�����0. 1mol��L ��1KI��Һ����0. 1mol��L ��1 FeCl3��Һ����FeCl2��Һ���������KSCN��Һ����CCl4��
ʵ�鲽�裺
��ȡ5mL 0. 1mol��L ��1 KI��Һ,�ٵμ�5~6�Σ�Լ0.2ml��0. 1mol��L��1FeCl3��Һ
�ڳ�ַ�Ӧ����Һ�ֳ�����
��ȡ����һ�ݣ��μ��Լ�CCl4��������һ��ʱ�䣬CCl4������Ϻ�ɫ��˵����Ӧ���ɵ⡣
����ȡһ�ݣ��μ��Լ� �����Լ���ţ���������Ϊ ����˵���÷�Ӧ��һ�����ȡ�
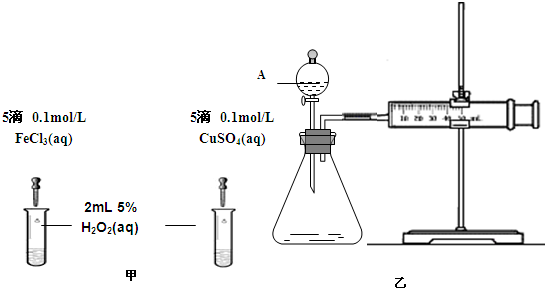
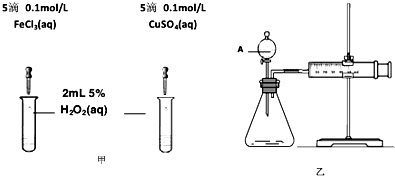
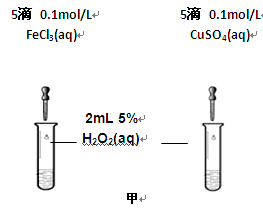
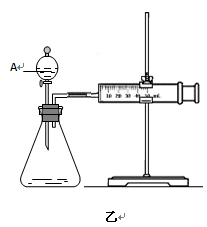
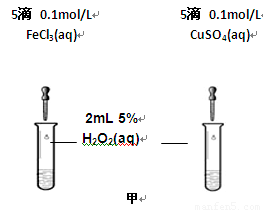
��2��Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�顣��ش�������⣺
�ٶ��Է�������ͼ��ͨ���۲� �����������ԱȽϵó����ۡ���ͬѧ�����FeCl3��Һ��ΪFe3+Ũ����ͬ��Fe2(SO4)3��Һ��Ϊ�������������� ��
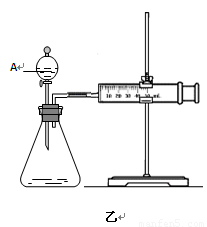
�ڶ�����������ͼ����ʾ��ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ʵ����һ����Ҫ������������ ������ţ���
���ռ�40ml��������Ҫ��ʱ�� ��CuSO4��Һ��FeCl3��Һ��Ũ�� �۹���������Һ��Ũ��
��1���Լ� �� ��2�֣������Լ���ţ� ������Һ��Ѫ��ɫ ��2�֣�
��2�����۲������Թ����������ɵ��ٶ���2�֣� ���ɣ��ų�Cl-��ʵ��ĸ�����2�֣�
�� �� ��2�֣�
����:���ʵ��̽��KI��Һ��FeCl3��Һ��Ӧ����һ�����ȡ��ֳ�������Һͨ����������KSCN��Һ�ĺ����Ե������жϡ�
Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч������ͬ��������Կ�����Ч�����۲������Թ����������ɵ��ٶ����ռ�40ml��������Ҫ��ʱ�� ��

 ������ҵ����ν�����������ϵ�д�
������ҵ����ν�����������ϵ�д�