��Ŀ����
ʳ���к���һ������þ���������ʣ��ӵ����е����ʧ��Ҫ�������ʡ�ˮ�֡������е������Լ����ա����ȶ�����ģ���֪��
�����ԣ�IO3-��Fe3����I2����ԭ�ԣ�S2O32-��I��
3I2��6OH��=5I����IO3-��3H2O��KI��I2  KI3
KI3
��1��ijѧϰС��Լӵ��ν���������ʵ�飺ȡһ�����ӵ���(���ܺ���KIO3��KI��Mg2����Fe3��)������������ˮ�ܽ⣬����ϡ�����ữ����������Һ��3�ݡ���һ����Һ�еμ�KSCN��Һ���Ժ�ɫ���ڶ�����Һ�м�����KI���壬��Һ��ʾ����ɫ����CCl4��ȡ���²���Һ���Ϻ�ɫ����������Һ�м�������KIO3����μӵ����Լ�����Һ����ɫ���� ��KSCN��Һ�Ժ�ɫ������Һ�к��е�������________(�����ӷ��ű�ʾ)��CCl4����ʾ�Ϻ�ɫ��������________(�õ���ʽ��ʾ)��
�� �ڶ�����Һ�м�������KI�����Ӧ�����ӷ���ʽΪ_______________________��
��2����I2����KI��Һ���ڵ��������£����Ƶ�KI3��H2O����������Ϊʳ�μӵ���Ƿ���ʣ�(��ǡ���)����˵�����ɣ�________________________________��
��3��Ϊ����ӵ���(����KI)���ȶ��ԣ��ɼ��ȶ������ٵ����ʧ�������������п�����Ϊ�ȶ�������________��
A��Na2S2O3 B��AlCl3 C��Na2CO3 D��NaNO2
��1���� Fe3+ 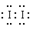 ��5I-+IO3��+6H+=3I2+3H2O��2Fe3����2I�D
��5I-+IO3��+6H+=3I2+3H2O��2Fe3����2I�D 2Fe2����I2��
2Fe2����I2��
��2�������Ʊ���KI3�����ֽ�ΪI2����I2������������
��3��AC
��������
�����������ij�ӵ��ο��ܺ���KIO3��KI��Mg2����Fe3����������ˮ�ܽ⣬����ϡ�����ữ����Һ��Ϊ3�ݡ��ӵ�һ����Һ�еμ�KSCN��Һ���Ժ�ɫ����֪�üӵ����к���Fe3������Ӧ�� Fe3��+ 3SCN-= Fe(SCN)3��Fe(SCN)��Ѫ��ɫ���ӵڶ�����Һ�м�����KI���壬��Һ�Ե���ɫ����CCl4��ȡ���²���Һ���Ϻ�ɫ����֪�е����ɡ�������Ϊ ���ڡ������ԣ�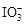 ��Fe3����I2���� ������KI��
��Fe3����I2���� ������KI��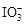 ��Fe3�����ܽ�I��������I2���ɴ�Ҳ����֪���üӵ�������KIO3����������Һ�м�������KIO3����μӵ����Լ�����Һ����ɫ���ɴ˿�֪�üӵ����в���KI�����ԣ��ٸüӵ�����Һ�м�KSCN��Һ�Ժ�ɫ��������Fe(SCN)3 �� CCl4�����Ϻ�ɫ��������I2 ������ʽ
��Fe3�����ܽ�I��������I2���ɴ�Ҳ����֪���üӵ�������KIO3����������Һ�м�������KIO3����μӵ����Լ�����Һ����ɫ���ɴ˿�֪�üӵ����в���KI�����ԣ��ٸüӵ�����Һ�м�KSCN��Һ�Ժ�ɫ��������Fe(SCN)3 �� CCl4�����Ϻ�ɫ��������I2 ������ʽ �ڵڶ�����Һ�м�������KI�����Ӧ�����ӷ���ʽΪ��
�ڵڶ�����Һ�м�������KI�����Ӧ�����ӷ���ʽΪ��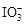 ��5I����6H��
��5I����6H�� 3I2��3H2O �� 2Fe3����2I�D
3I2��3H2O �� 2Fe3����2I�D 2Fe2����I2��
2Fe2����I2��
�Ƹ�����Ŀ��֪��KI3��H2O���ڵ��������£���I2����KI��Һ���Ƶá������������Ϣ����KI��I2 KI3���������Ƴ���KI3�ڳ����²��ȶ��ԣ����ȣ���ʪ���������ֽ�ΪKI��I2 ��KI���ױ������е�����������I2������������KI3��H2O��Ϊʳ�μӵ���Dz����ʵġ�
KI3���������Ƴ���KI3�ڳ����²��ȶ��ԣ����ȣ���ʪ���������ֽ�ΪKI��I2 ��KI���ױ������е�����������I2������������KI3��H2O��Ϊʳ�μӵ���Dz����ʵġ�
�� ��ӵ��Σ�����KI�����ȶ��ԣ���Ҫ�Ƿ�ֹI�D �����������������Ϣ����ԭ�ԣ�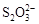 ��I������������ԭ��Ӧ��ǿ�����ɣ�����ѡNa2S2O3���ȶ��������������Ϣ��3I2��6OH��
��I������������ԭ��Ӧ��ǿ�����ɣ�����ѡNa2S2O3���ȶ��������������Ϣ��3I2��6OH��
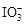 ��5I����3H2O������֪I2��OH�D�ᷢ����Ӧ����
��5I����3H2O������֪I2��OH�D�ᷢ����Ӧ����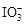 ��5I������Na2CO3ˮ��ʼ��ԣ����Ҳ������Na2CO3���ȶ�������ֹ�ӵ��Σ�����KI���ڳ�ʪ�����±���������������AlCl3��ˮ�������ԣ��һ�ԭ��I�� > Cl�D ,���������ȶ�����NaNO2����ǿ��ԭ������ʱ�ܱ��ֳ������ԣ���NaNO2��KI�ܷ���������ԭ��Ӧ��2NO2��+ 2I�D+ 4H��= 2NO��+ I2+2H2O ,�������ȶ�����
��5I������Na2CO3ˮ��ʼ��ԣ����Ҳ������Na2CO3���ȶ�������ֹ�ӵ��Σ�����KI���ڳ�ʪ�����±���������������AlCl3��ˮ�������ԣ��һ�ԭ��I�� > Cl�D ,���������ȶ�����NaNO2����ǿ��ԭ������ʱ�ܱ��ֳ������ԣ���NaNO2��KI�ܷ���������ԭ��Ӧ��2NO2��+ 2I�D+ 4H��= 2NO��+ I2+2H2O ,�������ȶ�����
���㣺�������ʵļ��顢������ԭ��Ӧ��������ԭ��Ӧ����ʽ�����ӷ�Ӧ�����ӷ�Ӧ����ʽ������ʽ���Լ�ʵ�鷽������ƵȻ�ѧ����֪ʶ�ͻ������ܡ�

 KI3
KI3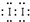

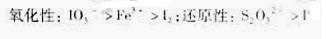
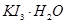 ����������Ϊʳ�μӵ���Ƿ���ʣ�
����������Ϊʳ�μӵ���Ƿ���ʣ�