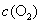��Ŀ����
��ҵ�ϲ���������������������������ж�����������װ������ͼ��ʾ����Ӧ����װ�е�ĵ�����Һ����������͵ⷢ���ķ�ӦΪ����������������ⷴӦ����
SO2+I2+2H2O==H2SO4+2HI
��1�����������뷴Ӧ�ܺ������������ӵ�ˮ���������________����д��ѧʽ���������
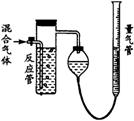 ��2����Ӧ������Һ��ɫ��ʧ��û�м�ʱֹͣͨ�������õĶ���������________���ƫ�ߡ���ƫ�͡�����Ӱ�족����
��2����Ӧ������Һ��ɫ��ʧ��û�м�ʱֹͣͨ�������õĶ���������________���ƫ�ߡ���ƫ�͡�����Ӱ�족����
��3����Ӧ���ڵĵ�ĵ�����ҺҲ������________����д�������ƣ����档
��4��������Һ���ΪVa mL��Ũ��Ϊc mol��L��1��N2��O2�����ΪVb mL��������Ϊ��״���µ����������c��Va��Vb��ʾ����������������Ϊ________��
��5��������װ�ø�Ϊ����ʵ��װ�ã��������⣬����ѡ�õ�����Ϊ________�������������ı�ţ���
a���ձ� b���Թ� c�����ƿ d������ƿ e����Ͳ f�������� g��˫����
��1��N2��O2 ��2��ƫ�� ��3�����Ը��������Һ
��4��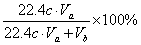 ��5��b��c��e��g��b��e��g��c��e��g���𰸲�Ψһ��
��5��b��c��e��g��b��e��g��c��e��g���𰸲�Ψһ��
[����]��1�����������뷴Ӧ���У�����SO2��I2������Ӧ��SO2+I2+2H2O==H2SO4+2HI�����������壬ʣ����������N2��O2�������������ӵ�ˮ���������N2��O2���������
��2����Ӧ������Һ��ɫ��ʧʱ��SO2�뷴Ӧ����������I2ǡ�÷�Ӧ����û�м�ʱֹͣͨ������δ��Ӧ��SO2����Ҳ�ŵ������ܣ�ʹN2��O2������ӣ����SO2�ĺ���ƫ�͡�
��3����ĵ�����Һ�������ǵ����е�I2��ȫ��SO2��ԭʱ����Һ����ɫ����ɫ����ɫ��ָʾ�յ�ģ���������SO2���巴Ӧ�����ʡ��ڴﵽ�յ�����ɫ�ı�Ŀɴ����ĵ�����Һ�������Ը��������Һ�ȡ�
��4����������Ӧ֪SO2��������ʵ������ڵ�����ʵ���������
Va��10��3 L��c mol��L��1=cVa��10��3 mol��
�������V (SO2)=cVa��10��3 mol��22.4 L��mol��1=22.4cVa mL��
����SO2�����������
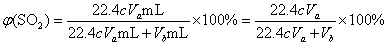 ��
��
��5��������װ�ÿ�֪��Ӧ�����ܱ����������ã���˿����Թܡ����ƿ���棬����ȡ��������ķ��������������⣬�������ɲ����������ų�Һ������ȷ�������ù��ƿ��˫��������Ͳ���������ܣ��ʸ�Ϊ����װ����ѡ����Ϊb��c��e��g��b��e��g��c��e��g��

��������������������Լ�����������ӦΪ2NO2(g)+O3(g) N2O5(g)+O2(g)������Ӧ�ں����ܱ������н��У������ɸ÷�Ӧ���ͼ���������ж���ȷ���ǣ� ��
N2O5(g)+O2(g)������Ӧ�ں����ܱ������н��У������ɸ÷�Ӧ���ͼ���������ж���ȷ���ǣ� ��
| A | B | C | D |
|
|
|
|
|
| �����¶ȣ�ƽ�ⳣ����С | 0~3s�ڣ���Ӧ����Ϊ |
| �ﵽƽ��ʱ�����ı� |
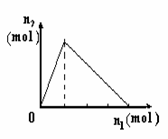
���� ��X����Y��Һ�У���
��X����Y��Һ�У��� �� �������ʵ���n2������X�����ʵ���n1�Ĺ�ϵ��ͼ��ʾ������ͼ��ʾ�������(����)
�� �������ʵ���n2������X�����ʵ���n1�Ĺ�ϵ��ͼ��ʾ������ͼ��ʾ�������(����)
| A | B | C | D | |
| X | NaOH | AlCl3 | HCl | NaAl |
| Y | AlCl3 | NaOH | NaAl | HCl |

 Pb
Pb ˮ������ӷ���ʽ��HCO
ˮ������ӷ���ʽ��HCO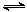 H2CO3+H3O+
H2CO3+H3O+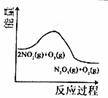
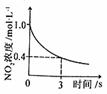


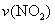 =0.2 mol��L-1
=0.2 mol��L-1 ʱ�����������ƽ�������ƶ�
ʱ�����������ƽ�������ƶ�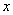 ����
����