��Ŀ����
��10�֣�ʵ������ȡ������������Ҫ�������£�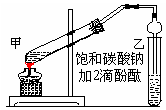
���ڼ��Թܣ���ͼ���м���2mLŨ���ᡢ3mL�Ҵ���2mL����Ļ����Һ��
�ڰ���ͼ���Ӻ�װ�ã�װ�����������ã���������Һ����С����ȵؼ���3��5min��
�۴��Թ����ռ���һ���������ֹͣ���ȣ������Թ�B��������Ȼ���ô��ֲ㡣�ܷ�������������㡢ϴ�ӡ����
��1����Ӧ��Ũ�����������___________________д����ȡ���������Ļ�ѧ����ʽ��___________________________________��
��2������ʵ���б���̼������Һ�������ǣ�����ĸ����_______________��
| A���к�������Ҵ��� |
| B���к����Ტ���ղ����Ҵ��� |
| C�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ������� |
| D�������������ɣ��������ʡ� |
��1����������ˮ����2�֣� CH3COOH+CH3CH2OH CH3COOCH2CH3+H2O ��2�֣� ��2��BC ��2�֣� ��3����Һ©�����Ͽڵ�.����2�֣�
CH3COOCH2CH3+H2O ��2�֣� ��2��BC ��2�֣� ��3����Һ©�����Ͽڵ�.����2�֣�
����

��ϰ��ϵ�д�
�����Ŀ
����ʵ������ȡ����������ʵ����������ͷ�Ӧԭ���������У�����ȷ���ǣ�������
| A�������ʯ��������Ϊ�˷�ֹҺ�屩�� | B�������¶Ȳ��˹�����Ϊ�˷�ֹ�Ҵ�������Ļӷ� | C��������ռ��ڱ���Na2CO3��Һ�Ϸ�����Ϊ�˳�ȥ���������е��Ҵ��ʹ��� | D����Ӧ�������������ԭ�ӣ�-H�����Ҵ����ǻ���-OH�� |
 ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ��
ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A�з���Ũ���ᣬB�з����Ҵ�����ˮ�����ƣ�D�з��б���̼������Һ�� ʵ������ȡ����������ʵ��װ����ͼ��ʾ����ش��������⣮
ʵ������ȡ����������ʵ��װ����ͼ��ʾ����ش��������⣮ ʵ��������ͼ��ʾװ����ȡ����������
ʵ��������ͼ��ʾװ����ȡ����������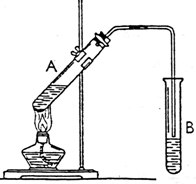 ��ͼΪʵ������ȡ����������װ��ͼ���Իش�
��ͼΪʵ������ȡ����������װ��ͼ���Իش�