��Ŀ����
��15�֣�����֪һ��̼ԭ�������������ǻ�ʱ����������ת�����������ͼ�ش����⣺
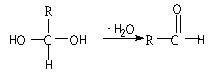
����֪F���������ص㣺���ܸ�FeCl3��Һ������ɫ��Ӧ����1mol F��������2mol NaOH����Fֻ��һ�����ұ����ϵ�һ�ȴ���ֻ�����֡�

��1����֪B����Է�������Ϊ178����ȼ�ղ�����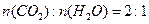 ����B�ķ���ʽ�� ������F���ܵĽṹ��ʽΪ�� ��
����B�ķ���ʽ�� ������F���ܵĽṹ��ʽΪ�� ��
��2��д���ٵķ�Ӧ����ʽ�� ��
д���۵ķ�Ӧ����ʽ�� ��
��3����F��ͬ���칹���У���������ķ����廯������ �֡�

����֪F���������ص㣺���ܸ�FeCl3��Һ������ɫ��Ӧ����1mol F��������2mol NaOH����Fֻ��һ�����ұ����ϵ�һ�ȴ���ֻ�����֡�

��1����֪B����Է�������Ϊ178����ȼ�ղ�����
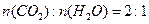 ����B�ķ���ʽ�� ������F���ܵĽṹ��ʽΪ�� ��
����B�ķ���ʽ�� ������F���ܵĽṹ��ʽΪ�� ����2��д���ٵķ�Ӧ����ʽ�� ��
д���۵ķ�Ӧ����ʽ�� ��
��3����F��ͬ���칹���У���������ķ����廯������ �֡�
��1��C10H10O3 (2��) 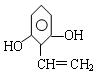 (2��)
(2��)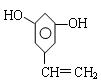 (2��)
(2��)
��2��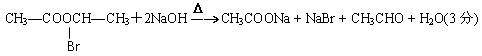
CH3CHO + 2Cu(OH)2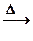 Cu2O
Cu2O + CH3COOH + 2H2O(3��)
+ CH3COOH + 2H2O(3��)
��3��6��(3��)
 (2��)
(2��) (2��)
(2��)��2��
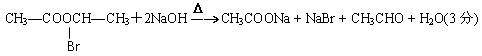
CH3CHO + 2Cu(OH)2
 Cu2O
Cu2O + CH3COOH + 2H2O(3��)
+ CH3COOH + 2H2O(3��)��3��6��(3��)
��

��ϰ��ϵ�д�
�����Ŀ
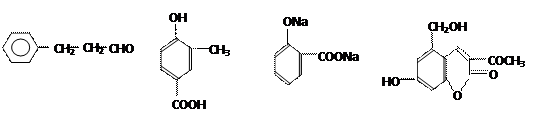
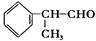 ��һ����Ҫ�Ļ���ԭ�ϣ���ϳ�·�����£�
��һ����Ҫ�Ļ���ԭ�ϣ���ϳ�·�����£�
 ��Ӧ�Ļ�ѧ����ʽΪ ��
��Ӧ�Ļ�ѧ����ʽΪ ��
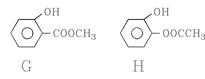 �� ��
�� ��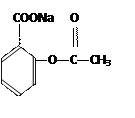
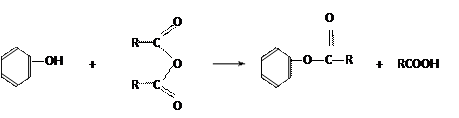

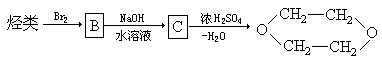
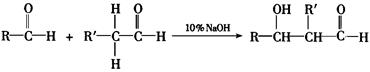
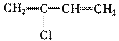 �ۺϵ�__________________________ _��
�ۺϵ�__________________________ _��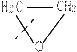 �����ߴ������ۺϵ�
�����ߴ������ۺϵ�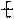 CH2CH2O
CH2CH2O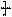 n����
n����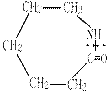 �����ۺϵ�_______________________________ ��
�����ۺϵ�_______________________________ ��