��Ŀ����
����Ŀ���л��������������а�������Ҫ�Ľ�ɫ���о��л��������Ҫ�����塣
�����ϡ���ϩ�ı���ģ������ͼ��ʾ��ʵ������ϩ��̼̼˫���ļ��ܣ�������ָ�Ͽ�1 mol��ѧ���������������615 kJ/mol��������̼̼�����ļ�����348 kJ/mol��ֻ��Ҫ���ٵ�����������ʹ˫���������һ�������ѣ� ������ϩ�Ļ�ѧ���ʽϻ��ã��������ӳɷ�Ӧ�ȵ�ԭ��������ϩ�ױ� �������������罫��ϩͨ������KMn04��Һ����Һ����ɫ��ȥ����ϩ������Ϊ������̼��

��.�ο��������ϣ������ѧ֪ʶ���ش��������⡣
��1����ϩ�Ľṹʽ��.
��2������˵���в���ȷ���� (����ţ�
A������ϩ��CH2=CHCl)������ԭ����ͬһ��ƽ����
B����ϡ�����е�̼̼˫����������ͬ��̼̼����
C����;����ˮ��ʱ���������Ը��������Һ����ˮ���ͷŵ���ϩ���Ա���ˮ������
D����ȥ�����е���ϩ������ѡ�����Ը��������Һ
��3��ʵ������ȡ����ϩ�л���SO2���������ʡ������Լ��У������ڼ�����ϩ�л��е�SO2���� (����ţ�
A����ˮ B�����Ը��������Һ C��Ʒ����Һ
��.��֪��ϩ�ܷ�������ת��
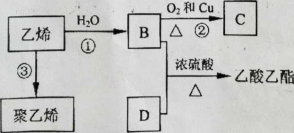
��1�� D�������������ŵ������� ��
��2����Ӧ����B��C)�Ļ�ѧ����ʽ�� ��
���𰸡�I����1�� ![]() ����2��b d��(3) c��II����1���Ȼ�����2��2CH3CH2OH+O2
����2��b d��(3) c��II����1���Ȼ�����2��2CH3CH2OH+O2 ![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��
��������
���������I.��1����ϩ�Ľṹ��ʽΪCH2=CH2����ṹʽΪ��![]() ����2��a����ϩ��ƽ��ṹ������ԭ�ӹ��棬����ϩ���Կ�������ԭ��ȡ����ϩ�ϵ���ԭ�ӣ�������ԭ�ӹ��棬��˵����ȷ��b��������Ŀ�е���Ϣ��̼̼˫���ļ��ܱȵ����ļ��ܴ������ϩ��̼̼˫������2��̼̼��������˵������c����ϩ��ʹ���Ը��������Һ��ɫ����ϩ�Ǵ���������Ը��������Һ������ϩ������ˮ�����죬��˵����ȷ��d����ϩ�����Ը��������Һ������CO2�������д����µ�����CO2����˵������(3)��ϩ��SO2������ˮ�����Ը��������Һ��ɫ����SO2��ʹƷ����Һ��ɫ������ϩ����ʹƷ����Һ��ɫ����ѡ��c��ȷ��II.��1��B��D������������B����ϩ��ˮ�����ӳɷ�Ӧ�������Ҵ�����DΪ���ᣬ����������Ȼ���(2)��Ӧ�����������������䷴Ӧ����ʽΪ��2CH3CH2OH+O2
����2��a����ϩ��ƽ��ṹ������ԭ�ӹ��棬����ϩ���Կ�������ԭ��ȡ����ϩ�ϵ���ԭ�ӣ�������ԭ�ӹ��棬��˵����ȷ��b��������Ŀ�е���Ϣ��̼̼˫���ļ��ܱȵ����ļ��ܴ������ϩ��̼̼˫������2��̼̼��������˵������c����ϩ��ʹ���Ը��������Һ��ɫ����ϩ�Ǵ���������Ը��������Һ������ϩ������ˮ�����죬��˵����ȷ��d����ϩ�����Ը��������Һ������CO2�������д����µ�����CO2����˵������(3)��ϩ��SO2������ˮ�����Ը��������Һ��ɫ����SO2��ʹƷ����Һ��ɫ������ϩ����ʹƷ����Һ��ɫ����ѡ��c��ȷ��II.��1��B��D������������B����ϩ��ˮ�����ӳɷ�Ӧ�������Ҵ�����DΪ���ᣬ����������Ȼ���(2)��Ӧ�����������������䷴Ӧ����ʽΪ��2CH3CH2OH+O2 ![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
