��Ŀ����
1840���˹����һϵ��ʵ����ʵ�ó����ɣ���ָ����������һ����Ӧ���Էֲ����У��������Ӧ�����ջ�ų��������ܺ��������Ӧһ�η���ʱ���ջ�ų���������ͬ��������18���ͷ��ֵ�һ����Ҫ���ɣ���Ϊ��˹���ɣ���֪1mol ���ʯ��ʯī�ֱ�����������ȫȼ��ʱ�ų�������Ϊ�����ʯ��395.41kJ��ʯī��393.51kJ������ʯת��ʯīʱ�����Ȼ������ȣ�______������ֵ��_______���ɴ˿������ȶ�����______����ȡ���ʯ��ʯī��Ͼ��干1mol ��O2����ȫȼ�գ���������ΪQkJ������ʯ��ʯī�����ʵ���֮��Ϊ______���ú�Q�Ĵ���ʽ��ʾ����
�ɸ�˹���ɿ�֪��Ҫ�õ����ʯת��Ϊʯī�������仯���ɽ���������������ȫȼ��ʱ�ų�������������ɣ�
��C�����ʯ���TC��ʯī����H=-395.41kJ/mol-��-393.51kJ/mol��=-1.90kJ/mol��
�����ʯת��Ϊʯī�ų�������˵��ʯī���������ͣ��Ƚ��ʯ�ȶ���
���ʯ��ʯī��Ͼ��干1mol ��O2����ȫȼ�գ���������ΪQkJ��
��ʮ�ֽ��淨
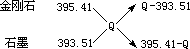
��
�ɵö������ʵ�����Ϊ
��
�ʴ�Ϊ�����ȣ�1.90kJ��ʯī��
��
��C�����ʯ���TC��ʯī����H=-395.41kJ/mol-��-393.51kJ/mol��=-1.90kJ/mol��
�����ʯת��Ϊʯī�ų�������˵��ʯī���������ͣ��Ƚ��ʯ�ȶ���
���ʯ��ʯī��Ͼ��干1mol ��O2����ȫȼ�գ���������ΪQkJ��
��ʮ�ֽ��淨

��
�ɵö������ʵ�����Ϊ
| Q-393.51 |
| 395.41-Q |
�ʴ�Ϊ�����ȣ�1.90kJ��ʯī��
| Q-393.51 |
| 395.41-Q |

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ