��Ŀ����
�����15�֣�
��4�������ȿ�����ͬ��ͬ��þ���Ͻ���Ʒ�١��ڡ��ۡ��ܡ��ס��ҡ�������4λͬѧ��ȡ1����Ʒ����������ʵ�飬�ⶨ�Ͻ���þ������������
52����ͬѧȡ��Ʒ��m1 g����������������Һ��Ӧ��Ȼ����ˣ�������Һ��ͨ������Ķ�����̼���壬�����ó������ˡ�ϴ�ӡ���ɡ����գ��õ�����������Ϊm1 g������Ʒ�ٺϽ���þ����������Ϊ_____________��
53����ͬѧȡ��Ʒ��m2 g�����������ᷴӦ��Ȼ��μӹ���������������Һ�����������ˡ�ϴ�ӡ���ɡ����գ��õ�����������Ϊm2 g������Ʒ�ںϽ���þ����������Ϊ____________��
54����ͬѧȡ��Ʒ��m3 g��������ϡ���ᷴӦ�����ֹ�����ȫ�ܽ⣬��״���µõ��������ΪV L������Ʒ����m3��ȡֵ��Χ��___________________________________________��
��ͬѧȡ��ͬ��������Ʒ�ֱܷ��30 mLͬŨ�ȵ����ᷴӦ����ȡ�Ͻ���������������������ת��Ϊ��״�������£�
| ʵ����� | a | b | c |
| �Ͻ�����/mg | 510 | 765 | 918 |
| �������/mL | 560 | 672 | 672 |
56��ͨ����������Ʒ���кϽ���þ������������
57����c��ʵ������������м���1.0 mol��L-1������������Һ���ٺ�������ʹʣ��Ͻ��е���ǡ����ȫ�ܽ⣿
52. 47.06%������2�֣�
53. 60%������2�֣�
54. 0.80V��m��1.07V
��2�֣�����д����27V/33.6��m��24V/22.4��9V/11.2��m��3V/2.8�����֣�
55. �������ᷴӦ��ȫ��n(H2)="0.672/22.4" ="0.03" mol����1�֣�
��c(HCl)="0.03��2/0.03=2.0" mol��L-1��1�֣�
56. ��Ͻ���Mg��Al�����ʵ����ֱ�Ϊx��y�����У�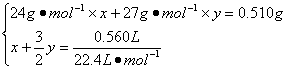
��1�֣�
��ã�x =" y" =" 0.01" mol��1�֣�
��w(Mg)="[(0.01" mol��24 g��mol-1)/0.510 g]��100%=47.06%��1�֣�
57. ����HCl ~ NaCl��Al ~ NaAlO2��֪
n(NaCl)="n(HCl)=2.0" mol��L-1��0.030 L="0.06" mol��1�֣�
n(NaAlO2)="n(Al)=0.01" mol��918/510="0.018" mol��1�֣�
����Na+�غ�ã�n(NaOH)="0.06" mol+0.018 mol="0.078" mol��1�֣�
��V(NaOH)="0.078" mol/1.0 mol��L-1="0.078" L="78" mL ��1�֣�
��c��ʵ��������78 mL������������Һ������ʹʣ��Ͻ��е���ǡ����ȫ�ܽ⡣
�������������52.þ���Ͻ��������������Ʒ�Ӧ���ᆳ��������̼��Ӧ���ó������ˡ�ϴ�ӡ���ɡ����գ��õ�����Ϊ����������ӦǰΪþ����Ӧ��Ϊ���������������䣬����Ԫ���غ��֪����������������������Ϊþ������������Ϊ16��3/��16��3+27��2��= 47.06%��53.�÷�Ӧ�õ��Ĺ���Ϊ����þ������Ԫ�������غ㣬����þ��þԪ�ص�������������þ���Ͻ���þԪ�ص�����������Ϊ24/��24+16��= 60%��
54.þ����ϡ���ᷴӦ���ĵ����ͬ������������Ҳ��ͬ���ʲ��ü�ֵ��ȷ���䷶Χ������ȫΪþʱ
��������ΪV /22.4��24=1.07V������ȫΪ��ʱV /22.4��2/3��27=0.80V,��Χ0.80V��m��1.07V��
55.��ͬѧʵ���������������̶���bc�����ᷴӦ��ȫ��a�������Ӧ��ȫ���ʼ��������Ũ�ȸ���bc����������ֱ�ӵó����������ᷴӦ��ȫ��n(H2)="0.672/22.4" ="0.03" mol��
��c(HCl)="0.03��2/0.03=2.0" mol��L-1
56.����a�������з���ʽ�ɽ⣬�ⷨ���𰸣�57.����Ԫ���غ㣬�Ͻ��������ձ�ΪNaAlO2����ʼ������������ձ�Ϊ�Ȼ��ƣ��˿ɳ�Ϊ��̬��������
n(NaCl)="n(HCl)=2.0" mol��L-1��0.030 L="0.06" mol����ԭ���غ㣩
n(NaAlO2)="n(Al)=0.01" mol��918/510="0.018" mol����ԭ���غ㣩
����Na+�غ�ã�n(NaOH)="0.06" mol+0.018 mol="0.078" mol
��V(NaOH)="0.078" mol/1.0 mol��L-1="0.078" L="78" mL ��1�֣�
��c��ʵ��������78 mL������������Һ������ʹʣ��Ͻ��е���ǡ����ȫ�ܽ⡣
���㣺���黯ѧ�����й����⡣

 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д����ǻ��õļ����Ԫ�أ��Ƽ��仯�������������������й㷺��Ӧ�á�
������м��㣺
��1���������ƣ�NaN3����ײ���ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ҡ���78�˵���������ȫ�ֽ⣬������״���µ���___________________L ��
��2����-�غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05 g��-�غϽ�����200 mLˮ����0.075 mol������������Һ���������Ƶ����ʵ���Ũ��______________________������Һ������仯����
��3������������Һ�����������ˣ��õ��������Ƶ���Һ�������Һ��ͨ�������̼�������з�Ӧ�� 2NaAl(OH)4+CO2��2Al(OH)3��+Na2CO3+H2O����֪ͨ�������̼112 L����״���£������ɵ�Al(OH)3��Na2CO3�����ʵ���֮��Ϊ4:5���������Һ��ͨ��Ķ�����̼Ϊ224L����״���£����������ɵ� Al(OH)3��Na2CO3�����ʵ��������ֵ��
��4��Ϊ�ⶨij�������պ�������������������е�Ԫ�ص������������ֽ���ͬ��������ι���ֱ���뵽50.00mL��ͬŨ�ȵ�����������Һ�У���ˮԡ����������ȫ���ݳ�(���¶�����β��ֽ�)�������徭�������Ũ����������ȫ���ⶨŨ�������ӵ����������ֲⶨ������±���
| ������/g | 10.00 | 20.00 | 30.00 | 50.00 |
| Ũ�������ӵ�����/g | m | m | 1.29 | 0 |
�Իش�
����εijɷ�Ϊ_______________________________��
������е�Ԫ�ص���������Ϊ��_______________________________(����ʽ���㣩��
ij�о���ѧϰС���������ռ���������Ϣ���ء��ơ��ơ�þ�Ȼ��ý���������CO2������ȼ�ա����Ƕ�����CO2������ȼ�պ�IJ����еİ�ɫ���ʽ���������̽����
ʵ�飺��ȼ�յ���Ѹ�����뵽ʢ��CO2�ļ���ƿ�У��������м���ȼ�գ���Ӧ����ȴ��ƿ���ź�ɫ������ƿ����𤸽�Ű�ɫ���ʡ�
�������
����1����ɫ������Na2O��
����2����ɫ������Na2CO3��
����3����ɫ������Na2O��Na2CO3�Ļ���
���ʵ�鷽����֤����
��С���ȼ�պ�İ�ɫ����
��������̽����
| ʵ�鷽�� | ʵ����� | ʵ������ | ���� |
| ����1 | ȡ������ɫ�������Թ��У���������ˮ������Ʒȫ������ˮ�������м�����ɫ��̪��Һ | ��Һ��ɺ�ɫ | ��ɫ����ΪNa2O |
| ����2 | ��ȡ������ɫ�������Թ��У���������ˮ������Ʒȫ������ˮ�������м��������CaCl2��Һ | ���ְ�ɫ���� | |
| | �ھ���Ƭ�̣�ȡ�ϲ���Һ���Թ��У��μ���ɫ��̪��Һ | ���������� |
��˼���뽻����
(1)��ͬѧ��Ϊ����1�õ��Ľ��۲���ȷ���������� ��
(2)��ͬѧ��Ϊ����2�õ��Ľ�����ȷ����ɫ����Ϊ ��
(3)ͨ������1�ͷ���2��ʵ�飬����Ϊ�������������У� ��������������� ��
(4)���ڶ�����̼��ȼ�յĻ�ѧ����ʽΪ ��
(5)��ͬѧ��Ϊ��ɫ�����п������������ơ����Ƿ�ͬ���ͬѧ�Ĺ۵㣿���������ɣ� ��
 Na3AlH6 (s)+
Na3AlH6 (s)+  Al (s) + H2(g) ��H��+ 37 kJ��mol��1
Al (s) + H2(g) ��H��+ 37 kJ��mol��1 H2(g) ��H��+ 70.5 kJ��mol��1
H2(g) ��H��+ 70.5 kJ��mol��1