��Ŀ����
��6�֣��ϳ�����ѹ�����º����10m3�״��ϳ������ڴ��������£����м״��ϳɣ���Ҫ��Ӧ�ǣ�2H2(g)
+ CO(g)  CH3OH(g)
��H =-181.6kJ��mol-1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OH(g)
��H =-181.6kJ��mol-1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
|
���� |
H2 |
CO |
CH3OH |
|
Ũ��/��mol��L��1�� |
0.2 |
0.1 |
0.4 |
�� �Ƚϴ�ʱ�����淴Ӧ���ʵĴ�С��v�� v�� ���>������<������)��
�� ������ͬ�����CO��H2����T5�淴Ӧ��10 min��ﵽƽ�⣬��ʱc(H2)��0.4 mol��L��1�����ʱ���ڷ�Ӧ����v(CH3OH) �� mol��(L��min)��1��
�� ���������У��ϳ���Ҫ����ѭ������Ŀ���� ��
�� > �� 0.03 mol��L��1��min��1 �� ���ԭ��CO��H2�������ʣ�����߲�����������ɣ���
����������1���������ʵ�Ũ�ȿ�֪����ʱ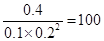 ��160�����Է�Ӧ������Ӧ������У�������Ӧ���ʴ����淴Ӧ���ʡ�
��160�����Է�Ӧ������Ӧ������У�������Ӧ���ʴ����淴Ӧ���ʡ�
��2�����ݣ�1����֪����ʼʱ������Ũ����1.0mol/L�������ڣ�2����������Ũ�ȱ仯����0.6mol/L�������ɼ״���0.3mol/L����״��ķ�Ӧ������0.3mol/L��10min��0.03 mol��L��1��min��1��
��3�����������У��ϳ�������ѭ�����������ԭ��CO��H2�������ʡ�

��ú����ת���ɺϳ�����Ȼ��ͨ��һ̼����·�ߺϳɸ�����Ʒ��ʯ����Ʒ��һ̼�����ļ�Ϊ��Ҫ�������й�����ǰ������δ���൱һ��ʱ�ڽ���Ϊһ̼��������Ҫ����
��ȥˮ�������ˮú����55��59%��H2��15��18%��CO��11��13%��CO2��������H2S��CH4����ȥH2S�ɲ��ô���Ǵ�ת����������CH4ת����CO���õ�CO��CO2��H2�Ļ�����壬������ĺϳɼ״�ԭ���������ɽ��м״��ϳɡ�
��1����ˮú������Ҫ��ѧ��Ӧ����ʽΪ��C��s��+H2O��g��CO��g��+H2��g�����˷�Ӧ��
���ȷ�Ӧ��
�� �˷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ�� ��
������������̼��ת���ʵĴ�ʩ�� ��
A������C��s�� B������H2O��g�� C�������¶� D������ѹǿ
��2����CH4ת����CO����ҵ�ϳ����ô�ת���������䷴Ӧԭ��Ϊ��
CH4 (g)+3/2O2 (g)CO (g)+2H2O (g) ��H=��519KJ/mol����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ��
�� X��T1��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
�� Y��T2��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
�� Z��T3��ʱ��Ч����ߣ���ʹ�淴Ӧ���ʼӿ�Լ1��106����
��֪��T1��T2��T3������������Ϣ������Ϊ��������Ӧ��ѡ������˴����� ���X����Y����Z������ѡ��������� ��
��3���ϳ�����ѹ�����º����10m3�״��ϳ������ڴ��������£����м״��ϳɣ���Ҫ��Ӧ���£�
2H2(g) + CO(g) CH3OH(g)����H ����90.8kJ��mol��1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
| ���� | H2 | CO | CH3OH |
| Ũ��/��mol��L��1�� | 0.2 | 0.1 | 0.4 |
�� �Ƚϴ�ʱ�����淴Ӧ���ʵĴ�С��v�� v�� ���>������<������)��
�� ������CO��H2����T5�淴Ӧ10min�ﵽƽ�⣬c(H2)��0.4 mol��L��1�����ʱ���ڷ�Ӧ����v(CH3OH) �� mol��1��(Lmin)��1��
��4�����������У��ϳ���Ҫ����ѭ������Ŀ���� ��
��6�֣��ϳ�����ѹ�����º����10m3�״��ϳ������ڴ��������£����м״��ϳɣ���Ҫ��Ӧ�ǣ�2H2(g) + CO(g)  CH3OH(g) ��H =-181.6kJ��mol-1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OH(g) ��H =-181.6kJ��mol-1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
| ���� | H2 | CO | CH3OH |
| Ũ��/��mol��L��1�� | 0.2 | 0.1 | 0.4 |
��������ͬ�����CO��H2����T5�淴Ӧ��10 min��ﵽƽ�⣬��ʱc(H2)��0.4 mol��L��1�����ʱ���ڷ�Ӧ����v(CH3OH) �� mol��(L��min)��1��
�����������У��ϳ���Ҫ����ѭ������Ŀ���� ��
��ú����ת���ɺϳ�����Ȼ��ͨ��һ̼����·�ߺϳɸ�����Ʒ��ʯ����Ʒ��һ̼�����ļ�Ϊ��Ҫ�������й�����ǰ������δ���൱һ��ʱ�ڽ���Ϊһ̼��������Ҫ����
��ȥˮ�������ˮú����55��59%��H2��15��18%��CO��11��13%��CO2��������H2S��CH4����ȥH2S�ɲ��ô���Ǵ�ת����������CH4ת����CO���õ�CO��CO2��H2�Ļ�����壬������ĺϳɼ״�ԭ���������ɽ��м״��ϳɡ�
��1����ˮú������Ҫ��ѧ��Ӧ����ʽΪ��C��s��+H2O��g�� CO��g��+H2��g�����˷�Ӧ��
CO��g��+H2��g�����˷�Ӧ��
���ȷ�Ӧ��
�� �˷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ�� ��
������������̼��ת���ʵĴ�ʩ�� ��
| A������C��s�� | B������H2O��g�� | C�������¶� | D������ѹǿ |
CH4 (g)+3/2O2 (g)
 CO (g)+2H2O (g) ��H=��519KJ/mol����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ��
CO (g)+2H2O (g) ��H=��519KJ/mol����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ���� X��T1��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
�� Y��T2��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
�� Z��T3��ʱ��Ч����ߣ���ʹ�淴Ӧ���ʼӿ�Լ1��106����
��֪��T1��T2��T3������������Ϣ������Ϊ��������Ӧ��ѡ������˴����� ���X����Y����Z������ѡ��������� ��
��3���ϳ�����ѹ�����º����10m3�״��ϳ������ڴ��������£����м״��ϳɣ���Ҫ��Ӧ���£�
2H2(g) + CO(g)
 CH3OH(g)����H ����90.8kJ��mol��1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OH(g)����H ����90.8kJ��mol��1��T4���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������м���CO��H2����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�| ���� | H2 | CO | CH3OH |
| Ũ��/��mol��L��1�� | 0.2 | 0.1 | 0.4 |
�� ������CO��H2����T5�淴Ӧ10min�ﵽƽ�⣬c(H2)��0.4 mol��L��1�����ʱ���ڷ�Ӧ����v(CH3OH) �� mol��1��(Lmin)��1��
��4�����������У��ϳ���Ҫ����ѭ������Ŀ���� ��
��15�֣���ˮú����ת���ɺϳ�����Ȼ��ϳɸ�����Ʒ��ʯ����Ʒ�ǻ����ļ�Ϊ��Ҫ������ȥˮ�������ˮú����Ҫ��H2��CO��CO2��������H2S��CH4��������ȥH2S�ɲ��ô���Ǵ�ת����������CH4ת����CO���õ�CO��CO2��H2�Ļ�����壬������ĺϳɼ״�ԭ������
��1����ˮú������Ҫ��ѧ��Ӧ����ʽΪ��C(s)+H2O(g) CO(g)+H2(g)���˷�Ӧ�����ȷ�Ӧ��
�ٴ˷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ ��
������������̼��ת���ʵĴ�ʩ�� ��
A������C��s�� B������H2O��g�� C�������¶� D������ѹǿ
��2����CH4ת����CO����ҵ�ϳ����ô�ת���������䷴Ӧԭ��Ϊ��
 CH4(g)+3/2O2(g)
CO(g)+2H2O(g) ��H=-519kJ/mol����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ��
CH4(g)+3/2O2(g)
CO(g)+2H2O(g) ��H=-519kJ/mol����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ��
��X��750��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
��Y��600��ʱ��Ч����ߣ���ʹ����Ӧ���ʼӿ�Լ3��105����
��Z��440��ʱ��Ч����ߣ���ʹ�淴Ӧ���ʼӿ�Լ1��106����
��֪����������Ϣ������Ϊ��������Ӧ��ѡ������˴����� ���X����Y����Z������ѡ��������� ��
��3���ϳ�����ѹ�����º����״��ϳ������ڴ��������£����м״��ϳɣ���Ҫ��Ӧ���£�
 2H2(g)+CO(g)
CH3OHg); ��H=-90.8kJ��mol-1��T���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������п�ʼֻ����CO��H2����Ӧ10min���ø���ֵ�Ũ�����£�
2H2(g)+CO(g)
CH3OHg); ��H=-90.8kJ��mol-1��T���´˷�Ӧ��ƽ�ⳣ��Ϊ160�����¶��£����ܱ������п�ʼֻ����CO��H2����Ӧ10min���ø���ֵ�Ũ�����£�
|
���� |
H2 |
CO |
CH3OH |
|
Ũ��/��mol��L-1�� |
0.2 |
0.1 |
0.4 |
�ٸ�ʱ����ڷ�Ӧ����v(H2)= mol-1��L-1��min-1��
�ڱȽϴ�ʱ�����淴Ӧ���ʵĴ�С��
 ���>������<����=����
���>������<����=����
��4�����������У��ϳ���Ҫ����ѭ������Ŀ���� ��