��Ŀ����
����ѡһ������ѧ�뼼����
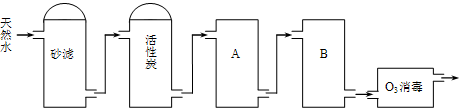
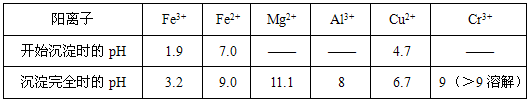
�ִ���п�ķ����ɷ�Ϊ��ʪ�������࣬������������п�ĸ���Ʒ�����ڸ߶��Խ������Իش�����������⡣
(1)����п�ǽ���п��(��Ҫ��ZnS)ͨ����ѡ������ʹ��ת��Ϊ����п���ٰ�����п�ͽ�̿��ϣ��ڹķ�¯�м�����1373��1573K��ʹп�����������Ҫ��ӦΪ2ZnS+3O2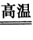 2ZnO+ 2SO2���ķ�¯�У�2C+O2
2ZnO+ 2SO2���ķ�¯�У�2C+O2 2CO��ZnO+CO
2CO��ZnO+CO Zn+CO2���û���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о����������¡�ʵ�����漰����ȣ�ÿ����Һ�к�����������������Ľ����ʵĹ�ϵ��ͼ�ף�������Һ���������������ȶ��������ʵ�Ӱ����ͼ�ҡ�
Zn+CO2���û���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о����������¡�ʵ�����漰����ȣ�ÿ����Һ�к�����������������Ľ����ʵĹ�ϵ��ͼ�ף�������Һ���������������ȶ��������ʵ�Ӱ����ͼ�ҡ�
�ִ���п�ķ����ɷ�Ϊ��ʪ�������࣬������������п�ĸ���Ʒ�����ڸ߶��Խ������Իش�����������⡣
(1)����п�ǽ���п��(��Ҫ��ZnS)ͨ����ѡ������ʹ��ת��Ϊ����п���ٰ�����п�ͽ�̿��ϣ��ڹķ�¯�м�����1373��1573K��ʹп�����������Ҫ��ӦΪ2ZnS+3O2
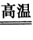 2ZnO+ 2SO2���ķ�¯�У�2C+O2
2ZnO+ 2SO2���ķ�¯�У�2C+O2 2CO��ZnO+CO
2CO��ZnO+CO Zn+CO2���û���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о����������¡�ʵ�����漰����ȣ�ÿ����Һ�к�����������������Ľ����ʵĹ�ϵ��ͼ�ף�������Һ���������������ȶ��������ʵ�Ӱ����ͼ�ҡ�
Zn+CO2���û���п�����IJ����к����ֽ������ʼ�In2O3������������ȡ����ij�о������Դ��о����������¡�ʵ�����漰����ȣ�ÿ����Һ�к�����������������Ľ����ʵĹ�ϵ��ͼ�ף�������Һ���������������ȶ��������ʵ�Ӱ����ͼ�ҡ� 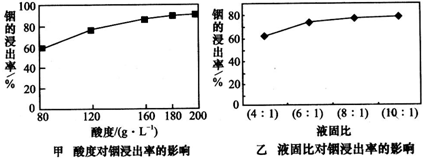
�ٵ����Ϊ196ʱ�������ʵ���Ũ��Ϊ____________��
�ڴӽ�Լԭ�Ϻͽ����ʿ��ǣ����˵���Ⱥ�Һ�̱ȷֱ�Ϊ��_________��________
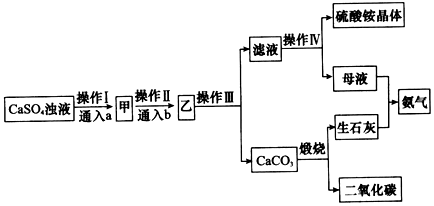
(2)ʪ����п����Ҫ��������Ϊ��
�ڴӽ�Լԭ�Ϻͽ����ʿ��ǣ����˵���Ⱥ�Һ�̱ȷֱ�Ϊ��_________��________
(2)ʪ����п����Ҫ��������Ϊ��

�ٴӱ��������ͳ������ԭ�ϽǶȣ���δ���������������_________________
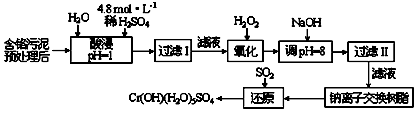
�ڳ�ȥ�����Һ�е���������H2O2�������ٵ���pHʹ֮�γ�Fe(OH)3������д��H2O2����
Fe2+�����ӷ���ʽ��______________
(3)�����Һ������Cd2+��Ϊ�˷�ֹ����Ⱦ�������ӣ������������ʵIJ��죬��������������Һ���룬��֪Zn(OH)2����������һ��Ҳ�������ԣ���д����������ӷ���ʽ��________________________��_________________��
�ڳ�ȥ�����Һ�е���������H2O2�������ٵ���pHʹ֮�γ�Fe(OH)3������д��H2O2����
Fe2+�����ӷ���ʽ��______________
(3)�����Һ������Cd2+��Ϊ�˷�ֹ����Ⱦ�������ӣ������������ʵIJ��죬��������������Һ���룬��֪Zn(OH)2����������һ��Ҳ�������ԣ���д����������ӷ���ʽ��________________________��_________________��
(1)��2mol��L-1����180��6:1
(2)�ٽ�����������������������ȣ���2Fe2++H2O2+ 2H+=2Fe3++2H2O
(3)Cd2++2OH-=Cd(OH)2����Zn2++4OH-=ZnO22-+ 2H2O{��Zn2++4OH-=[Zn(OH)4]2-}

��ϰ��ϵ�д�
 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
�����Ŀ
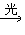 CH3CH2Cl��HCl
CH3CH2Cl��HCl 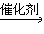 CH3CH2Cl
CH3CH2Cl