��Ŀ����
��17�֣��ɽ�������PCL�Ľṹ�ɱ�ʾΪ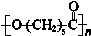 ����ϳ�·�����¡�
����ϳ�·�����¡�
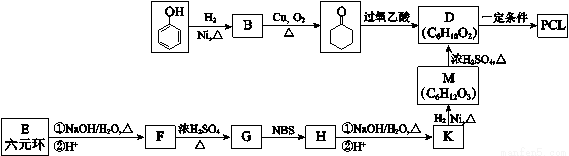
��֪��
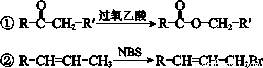
�ش��������⣺
��1��B�����������ŵ�������______��
��2��D�Ľṹ��ʽ��______��
��3��M�ķ��ӽṹ����֧����M����D�ķ�Ӧ������______��
��4��E��D��ͬ���칹�壬������ͬ�Ĺ����š�E�Ľṹ��ʽ��______��
��5��F����G�Ļ�ѧ����ʽ��______��
��6������˵����ȷ����______��
a��Kû��˳���칹
b��M��һ�������¿ɷ����ۺϷ�Ӧ
c��PCL�Ľṹ�к�������
��7��H����������������Һ��Ӧ�Ļ�ѧ����ʽ��______��
��8��M���������Z�����Ǻϳ�������ԭ��֮һ����B��ԭ�Ͽ��Ƶü����ᣬ���������Ϣ�����ϳ�·������ͼ�����Լ���ѡ����______��
��֪��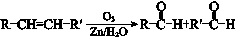
�ϳ�·������ͼʾ����
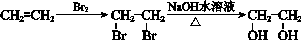
������Ʒ����Ҫ�ɷּ�����;��Ӧ����ȷ����
A | B | C | D | |
��Ʒ |
|
|
|
|
��Ҫ�ɷ� | (NH4)2SO4 | NaHCO3 | Si | Fe2O3 |
��; | ���� | ����� | ���� | Ϳ�� |
(15��)�������[KMnO4]�dz��õ�����������ҵ�������̿�(��Ҫ�ɷ���MnO2)Ϊԭ���Ʊ�������ؾ��塣�м����Ϊ�����[K2MnO4]����ͼ��ʵ����ģ���Ʊ��IJ������̣�
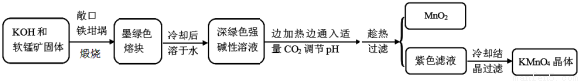
������ϣ�
�������ܽ��
���� | KMnO4 | K2CO3 | KHCO3��] | K2SO4 | CH3COOK |
20���ܽ�� | 6.4 | 111 | 33.7 | 11.1 | 217 |
�������[K2MnO4]
�����״��ī��ɫ�ᾧ����ˮ��Һ������ɫ�����������(MnO42��)��������ɫ��
��ѧ���ʣ���ǿ������Һ���ȶ��������ԡ����Ժ������Ի����£�MnO42���ᷢ���绯��Ӧ��
�Իش��������⣺
��1���������̿��KOH����ʱ��������ʯӢ������ѡ����������������______________��
ʵ���������������ձ�¶�ڿ����еĹ����������Ӧ�Ļ�ѧ����ʽΪ_______��
��2��ʵ��ʱ����CO2����������KHCO3�����µõ���KMnO4��Ʒ�Ĵ��Ƚ��͡���д��ʵ����ͨ������CO2ʱ��ϵ�п��ܷ�����Ӧ���ӷ���ʽ �� ��
����������ԭ��Ӧ���������ͻ�ԭ����������Ϊ_________________________��
��3������CO2��ͨ�������ѿ��ƣ���˶�����ʵ�鷽�������˸Ľ�������ʵ����ͨCO2��Ϊ���������ᡣ�������Ϸ�����ѡ����������________ ���õ��IJ�Ʒ���ȸ��ߡ�
A������ B��Ũ���� C��ϡ����
��4����ҵ��һ����ö��Ե缫����������Һ��ȡ������أ���д���õ�ⷴӦ�Ļ�ѧ����Ϊ_________��
��ͳ���ղ�����Ĥ��ⷨ���ڸ���Ӧ������MnԪ�������ʺ͵���Ч�ʶ���ƫ�͡���ͬѧ���뵽���ӽ���Ĥ����ⱥ��ʳ��ˮ����Ľ����������������ӽ���Ĥ�ָ����������е��(��ͼ)��
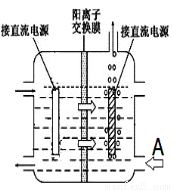
ͼ��A�ڼ������Һ���Ϊ__________��
ʹ�������ӽ���Ĥ�������MnԪ�������ʵ�ԭ��Ϊ_______________��
һ�������£�ͨ�����з�Ӧ��ʵ��ȼú��������Ļ��գ�����Y�ǵ��ʡ�
SO 2(g) + 2CO(g)  2X(g)+Y(l)
2X(g)+Y(l)
Ϊ�˲ⶨ��ij�ִ��������µķ�Ӧ���ʣ���ij�¶����ó��������崫������ò�ͬʱ���SO2��COŨ�����±���
ʱ��/s | 0 | 1 | 2 | 3 | 4 |
c(SO2)/mol��L-1 | 1.00 | 0.50 | 0.23 | 3.00��10-37 | 3.00��10-37 |
c(CO)/mol��L-1 | 4.00 | 3.00 | 2.46 | 2.00 | 2.00 |
����˵������ȷ����
A��X�Ļ�ѧʽΪCO2
B��ǰ1s��v(X)= 1.00 mol��L-1��s-1
C���û���ԭ��������SO 2�Ļ�ԭ��
D�����¶��£��˷�Ӧ��ƽ�ⳣ������ֵ��3.33��1036






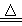 Na2O2
Na2O2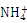
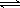 NH3��H2O+ H+
NH3��H2O+ H+