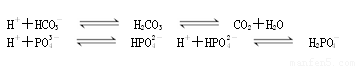��Ŀ����
����ѪҺ������pHԼΪ7.35��7.45�������������������ͻȻ�����ı�ͻ��������ж������ж���������������Σ�ա�����������Һ�Ļ���ϵͳ�д�������ƽ�⣺H++![]()
![]() H2CO3
H2CO3![]() CO2+H2O
CO2+H2O
H++![]()
![]()
![]()
H++![]()
![]()
![]()
����ά��ѪҺpH������ȶ����Ա�֤������������Լ�Ҫ�ش��������⣺
��1����ǿ�������ʽ����������Һ����������ϵͳ����ά����Һ�����ƽ�⣿_____________________________________________________________________
��2����ǿ�������ʽ����������Һ����������ϵͳ����������Һ�����ƽ�⣿_____________________________________________________________________
��3��ij�����ھ�����ע����������ˮ��ѪҺ�е�pH�Ƿ�ᷢ����ı仯��_____________________________________________________________________
��1������c(H+)����ƽ����ϵ���ƣ��Ӷ�ά��pH�ȶ���
��2������c(OH-)����ƽ����ϵ���ƣ��Ӷ�ά��pH�ȶ���
��3������ϵͳҲ�ֿܵ�ϡ�Ͷ���ҺpH��ɵ�Ӱ�죬���Բ���ʹѪҺpH������ı仯��

����ѪҺ������pHԼΪ7.35��7.45�������������������ͻȻ�����ı�ͻ��������ж������ж���������������Σ�ա�����������Һ�Ļ���ϵͳ�д�������ƽ�⣺
H++HCO3�� H2CO3
H2CO3 CO2+H2O
CO2+H2O
H++PO43�� HPO42��
HPO42��
H++HPO42�� H2PO4��
H2PO4��
����ά��ѪҺpH������ȶ����Ա�֤���������������˵���в���������
| A����ǿ�������ʽ����������Һ����������ϵͳ��ƽ�������ƣ��Ӷ�ά��pH�ȶ� |
| B����ǿ�������ʽ����������Һ����������ϵͳ��ƽ�������ƣ��Ӷ�ά��pH�ȶ� |
| C��ij�����ھ�����ע�Ĵ���������ˮ��ѪҺ��ϡ�ͣ��ᵼ��c(H+)�������٣�pHֵ���������ܻ�������ж� |
| D����������к����ʱ����CO2����ѪҺ����ʹƽ�������ƶ���c(H+)����pHֵ�Լ�С |
 H2CO3
H2CO3