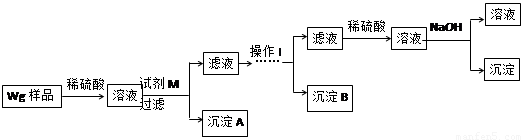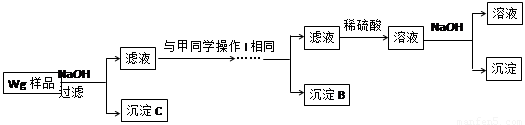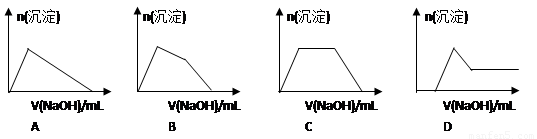��Ŀ����
ij��Ͻ�����ĩ����Mg�⣬������Al��Zn�е�һ�ֻ����֣���������
10%���ϡ�ij�о�С�����ʵ��̽���û�Ͻ�����ĩ������пԪ�صĴ��ڡ�
�����Լ�����Ʒ��pH��ֽ��ϡH2SO4��NaOH��Һ��ϡNH3��H2O��
��С��̽���������£�
�� �������ϣ�
| ��þ������п��������ɫ�Ľ��� ��п(Zn)������NaOH��Һ��Ӧ����H2 ��Zn(OH)2Ϊ��ɫ���壬������ˮ��������ǿ�NH3��H2O ��Zn2+���γ���������Zn(NH3)4��2+�����������ǿ��ֽ�����Zn2+��NH4+ |
�� ������裺
��1������٣��û�Ͻ�����ĩ�г�þ�����________Ԫ��
����ڣ��û�Ͻ�����ĩ�г�þ�����________Ԫ��
����ۣ��û�Ͻ�����ĩ�г�þ���������пԪ��
�� ʵ��̽����
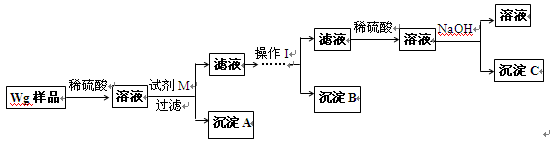
��ͬѧ���ڼ�������ʵ�鷽�����£�
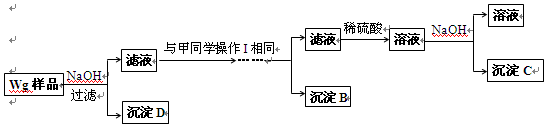
��ͬѧͬ�����ڼ���3�����һʵ�鷽�����£�
��2���Լ�M��________________������B�� ��
��3����ͬѧ��Ϊ��ͬѧ�ķ����ȼ�ͬѧ�ĺã������� ��
��4����ͬѧ�о��˼ס�����ͬѧ�ķ�����������һ�ַ����Ļ����������㷽���ⶨ��Wg��Ʒ�н���þ(Mg)�����������ķ����� ��
��5�����������Ҫ�����ǣ�����Һ����μ��� ��ֱ�����ɵij����պ��ܽ⣬�ټ��������� ��
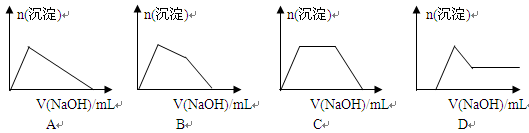
��6��ij��Һ�к���Zn2+��Al3+��NH4+��SO42�������ӣ���������μ���NaOH��Һ��
�����ɳ��������ʵ��������NaOH��Һ�����ϵ��ͼ����ȷ����__________��
��1������٣�������Al����1�֣���
����ڣ�п����Zn����1�֣����������տɵ�����
��2��NaOH��Һ��������������Һ���ޡ���Һ�����ֲ��۷֡�2�֣���
Al(OH)3 (2��)
��3�������1�֣�����Լ�Լ���1�֣���
��4��������Cϴ�ӡ�С�ĸ���������2�֣���
��5��ϡ���ᣨ2�֣���ϡ��ˮ��2�֣��� ��6��B ��2�֣�
����:��

��������ϣ�
| ��þ������п��������ɫ�Ľ��� ��п��Zn��������NaOH��Һ��Ӧ����H2 ��Zn��OH��2Ϊ��ɫ���壬������ˮ��������ǿ�NH3?H2O ��Zn2+���γ��������[Zn��NH3��4]2+�����������ǿ��ֽ�����Zn2+��NH4+��������裺 ��1������٣��û�Ͻ�����ĩ�г�þ����� Al Al Ԫ������ڣ��û�Ͻ�����ĩ�г�þ����� Zn Zn Ԫ������ۣ��û�Ͻ�����ĩ�г�þ���������пԪ�� ��ʵ��̽���� ��ͬѧ���ڼ�������ʵ�鷽�����£���ͬѧͬ�����ڼ���������һʵ�鷽�����£� 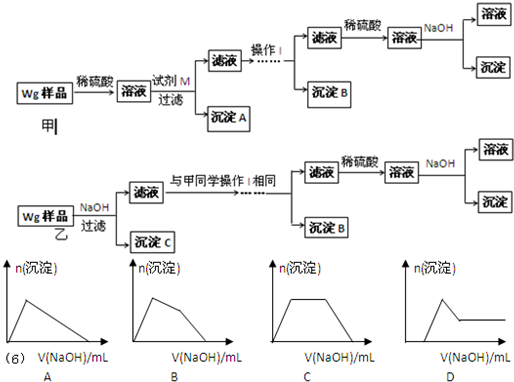 ��2���Լ�M�� NaOH��Һ NaOH��Һ ������B��Al��OH��3 Al��OH��3 ����3����ͬѧ��Ϊ��ͬѧ�ķ����ȼ�ͬѧ�ĺã������� �������Լ�Լ� �������Լ�Լ� ����4����ͬѧ�о��˼ס�����ͬѧ�ķ�����������һ�ַ����Ļ����������㷽���ⶨ��Wg��Ʒ�н���þ��Mg�������������ķ����� ������Cϴ�ӡ�С�ĸ������� ������Cϴ�ӡ�С�ĸ������� ����5�����������Ҫ�����ǣ�����Һ����μ��� ϡ���� ϡ���� ��ֱ�����ɵij����պ��ܽ⣬�ټ���������ϡ��ˮ ϡ��ˮ ����6��ij��Һ�к���Zn2+��Al3+��NH4+��SO42-�����ӣ���������μ���NaOH��Һ�������ɳ��������ʵ��������NaOH��Һ�����ϵ��ͼ����ȷ���� B B ��
ij��Ͻ�����ĩ����Mg�����Al��Zn�е�һ�ֻ����֣���������10%���ϡ�ij�о�С�����ʵ��̽���û�Ͻ�����ĩ������пԪ�صĴ��ڡ� �����Լ�����Ʒ��pH��ֽ��ϡH2SO4��NaOH��Һ��ϡNH3��H2O�� ��С��̽���������£� ��������ϣ�
��������裺 ��1������٣��û�Ͻ�����ĩ�г�þ�������Ԫ�أ� ����ڣ��û�Ͻ�����ĩ�г�þ�����пԪ�أ� ����ۣ� �� ��ʵ��̽���� ��ͬѧ���ڼ�������ʵ�鷽�����£�
��ͬѧͬ�����ڼ���3�����һʵ�鷽�����£�
��2���Լ�M��________________������B�� �� ��3����ͬѧ��Ϊ��ͬѧ�ķ����ȼ�ͬѧ�ĺã������� �� ��4����ͬѧ�о��˼ס�����ͬѧ�ķ�����������һ�ַ����Ļ����������㷽���ⶨ��Wg��Ʒ�н���þ(Mg)�����������ķ����� �� ��5�����������Ҫ�����ǣ�����Һ����μ��� ��ֱ�����ɵij����պ��ܽ⣬�ټ��������� �� ��6��ij��Һ�к���Zn2+��Al3+��NH4+��SO42�������ӣ���������μ���NaOH��Һ�������ɳ��������ʵ��������NaOH��Һ�����ϵ��ͼ����ȷ����__________��
|