��Ŀ����
���û�ѧ��Ӧԭ���о������������е�ʵ���������ʮ����Ҫ�����壺
�������������ϳɰ��ǻ�ѧ��ҵ�м�Ϊ��Ҫ�ķ�Ӧ�����Ȼ�ѧ����ʽ�ɱ�ʾΪ��N2(g)��3H2(g)  2NH3(g)����H����92 kJ��mol��1����ش��������⣺
2NH3(g)����H����92 kJ��mol��1����ش��������⣺
(1)ȡ1 mol N2(g)��3 mol H2(g)����һ�ܱ������У��ڴ�������ʱ���з�Ӧ����÷�Ӧ�ų��������ߣߣߣߣ�92 kJ(����ڡ������ڡ���С�ڡ�)��ԭ���ǣߣߣߣߣߣߣߣ��������������H������(������С�����䡱)��
(2)��֪���ֱ��ƻ�1 mol N��N����1 mol H��H����Ҫ���յ�����Ϊ��946 kJ��436 kJ�����ƻ�1 mol N��H����Ҫ���յ�����Ϊ�ߣߣߣߣߣ�kJ��
(3)N2H4����Ϊ��NH3�����е�H����NH2ȡ���IJ������������N2H4(g)Ϊȼ�ϣ�NO2Ϊ����������N2��H2O(g)��
��֪��N2(g)��2O2(g)===2NO2(g) ��H1����67��7 kJ��mol��1
N2H4(g)��O2(g)===N2(g)��2H2O(g) ��H2����534 kJ��mol��1��
��1 mol N2H4��ȫ��Ӧ���Ȼ�ѧ����ʽΪ ��
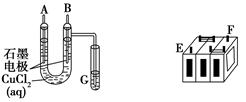
����ijǦ���ص�����������DZ�ĥ��������ͼװ�����ʵ�飬ʶ�����Ǧ���ص���������
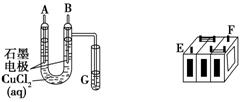
(1)��A��E��B��F����B�缫���� ����ӦʽΪ ����˵��FΪ������
��2����Ǧ���ع���ʱ���ŵ磩����E���ڵ缫�ĵ缫��ӦʽΪ�� �����ʱ�ü�����ӵ�Դ�� ��������
��3�����øõ�ص��Cu(NO3)2 ��Һ�����ⷽ��ʽΪ
����0.2mol���ӷ���ת�ƣ����������ĵ�PbO2�����ʵ����� ��Ҫ��CuSO4��Һ�ָ�ԭ���������������� ,����Ϊ
���� (1)С�ڡ����ڸ÷�Ӧ�ǿ��淴Ӧ����Ӧ����ȫ��ת��Ϊ��������䡡(2)391��(3)N2H4(g)��NO2(g)===3/2 N2(g)��2H2O(g)����H����567.85 kJ��mol��1
����(1)���ݡ�2Cl�D�D2e��====Cl2��
��2��Pb-2e����SO42����PbSO4 ����
��3�� 0.1
mol CuO 8g
0.1
mol CuO 8g
��������

 �����ܿ����ϵ�д�
�����ܿ����ϵ�д�
 2NH3(g)����H����92 kJ��mol��1����ش��������⣺
2NH3(g)����H����92 kJ��mol��1����ش��������⣺