��Ŀ����
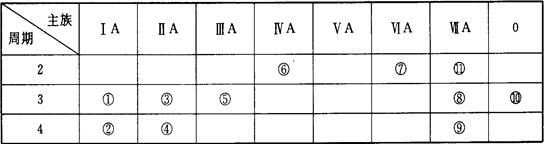
�±�ΪԪ�����ڱ��е�һ���֣��г���10��Ԫ����Ԫ�����ڱ��е�λ�á�����Ԫ�ط��š����ӷ��Ż�ѧʽ�ش��������⡣
| ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 | |
| ��2���� | �� | �� | ||||||
| ��3���� | �� | �� | �� | �� | �� | �� | ||
| ��4���� | �� | �� |
(1)10��Ԫ���У���ѧ��������õ��� ������Ԫ�ط��ţ�
��2���٢ۢ�����Ԫ������������Ӧˮ�����У�������ǿ���� �����ѧʽ���������ڲ����еĻ�ѧ�������� �� ��
��3��Ԫ�آߵ��⻯��Ļ�ѧʽΪ �����⻯�ﳣ���º�Ԫ�آڵĵ��ʷ�Ӧ�Ļ�ѧ����ʽ ��
��4���ۺ͢���Ԫ���γɻ�����ĵ���ʽ�� ���ٺ͢���Ԫ���γ��ĺ˻�����ĵ���ʽ�� ����������CO2��Ӧ�Ļ�ѧ����ʽΪ ��
(5)�ٺ͢�����������Ӧˮ�������Ӧ�����ӷ���ʽΪ��
��
(1) Ar
��2�� NaOH �� ���Ӽ��� ���Թ��ۼ�����ۼ����Լ����ԣ�
��3�� H2O ��2K + 2H2O =2KOH +H2�� ��
��4��![]() ��
�� 
2Na2O2 + 2CO2 = 2Na2CO3 +O2 ��
(5) Al(OH)3 + OH�� =AlO2�� + 2H2O

 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д��±�ΪԪ�����ڱ��е�һ���֣��г�10��Ԫ����Ԫ�����ڱ��е�λ�á�����Ԫ�ط��š����ӷ��Ż�ѧʽ�ش��������⡣
| | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | �� | | �� | | |
| 3 | �� | �� | �� | | �� | | �� | �� |
| 4 | �� | �� | | | | | | |
��2���٢ۢ�����Ԫ������������Ӧˮ�����У�������ǿ���� �����ѧʽ��
��3��Ԫ�آߵ��⻯��Ļ�ѧʽΪ �����⻯�ﳣ���º�Ԫ�آڵĵ��ʷ�Ӧ�Ļ�ѧ����ʽ
��4���ٺ͢�Ԫ�ص�����������Ӧˮ����Ļ�ѧʽΪ �� ���ۺ͢���Ԫ���γɻ�����ĵ���ʽ ���ٺ͢���Ԫ���γ��ĺ˻�����ĵ���ʽ ����������CO2��Ӧ�Ļ�ѧ����ʽ ��
��5���ٺ͢�����������Ӧˮ�������Ӧ�����ӷ���ʽΪ