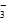��Ŀ����
�����£�0.1 mol��L��1��HA��Һ��c(OH��)/c(H��)��1��10��8��������������ȷ����
A��0.01 mol��L��1HA����Һ��c(H��)��1��10��4mol��L��1
B��pH��3��HA��Һ��pH��11��NaOH��Һ�������Ϻ�������Һ��c(Na��)��c(A��)��c(OH��)��c(H��)
C��Ũ�Ⱦ�Ϊ0.1 mol��L��1��HA��Һ��NaA��Һ�������Ϻ�������Һ�����ԣ���c(OH��)��c(H��)��c(HA)��c(A��)
D��pH��3��HA��Һ��pH��11��NaOH��Һ�������1?10��Ϻ�������Һ��c(OH��)��c(A��)��c(H��)��c(Na��)
A��0.01 mol��L��1HA����Һ��c(H��)��1��10��4mol��L��1
B��pH��3��HA��Һ��pH��11��NaOH��Һ�������Ϻ�������Һ��c(Na��)��c(A��)��c(OH��)��c(H��)
C��Ũ�Ⱦ�Ϊ0.1 mol��L��1��HA��Һ��NaA��Һ�������Ϻ�������Һ�����ԣ���c(OH��)��c(H��)��c(HA)��c(A��)
D��pH��3��HA��Һ��pH��11��NaOH��Һ�������1?10��Ϻ�������Һ��c(OH��)��c(A��)��c(H��)��c(Na��)
D
ˮ�����ӻ���c(OH��)��c(H��)��1��10��14�����0.1 mol��L��1��HA��Һ��c(OH��)/c(H��)��1��10��8���ɵ�c(OH��)��1��10��11mol/L,0.01 mol��L��1HA����Һ��c(H��)��1��10��4mol��L��1��A�����B�л�Ϻ�HA��������Һ�����ԣ�����C�и��ݵ���غ���c(H��)��c(A��)��ԭ���غ���2c(Na��)��c(A��)��c(HA)������c(A��)��c(Na��)��c(HA)��0��c(OH��)��c(H��)��c(Na��)��c(A��)��[c(HA)��c(A��)]/2��c(HA)��c(A��)��C����D���ǵ���غ�ʽ����ȷ��

��ϰ��ϵ�д�
 �������ϵ�д�
�������ϵ�д�
�����Ŀ
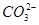 ��
��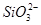 ��
��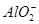 ��Cl��
��Cl��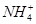 ��Na��
��Na��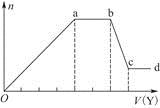
 Sn(OH)2
Sn(OH)2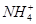 ��Cl����
��Cl����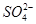 ��
��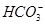 ��
��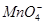 �еļ��֡�Ϊȷ����ɷ֣�������ʵ�飺��ȡ������Һ������������Na2O2���壬������ɫ��ζ������Ͱ�ɫ�������ټ���������NaOH��Һ���ɫ���������ܽ⣻����ȡ������Һ������HNO3�ữ��Ba(NO3)2��Һ���а�ɫ���������������ƶ���ȷ����
�еļ��֡�Ϊȷ����ɷ֣�������ʵ�飺��ȡ������Һ������������Na2O2���壬������ɫ��ζ������Ͱ�ɫ�������ټ���������NaOH��Һ���ɫ���������ܽ⣻����ȡ������Һ������HNO3�ữ��Ba(NO3)2��Һ���а�ɫ���������������ƶ���ȷ����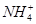 )/c(NH3��H2O)��ֵ����
)/c(NH3��H2O)��ֵ����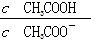 ��ֵ��С
��ֵ��С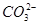 ˮ��̶ȼ�С����Һ��pH��С
ˮ��̶ȼ�С����Һ��pH��С ��2I����4H��
��2I����4H�� ��NO
��NO