题目内容
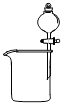
【题目】下图是某同学设计的木炭和浓硫酸反应并检验所得全部产物的实验装置.已知酸性高锰酸钾溶液具有强氧化性,可将S元素从+4价氧化为+6价.请回答: 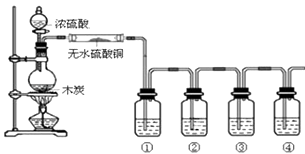
(1)装置中盛浓硫酸的仪器名称是;实验过程中大玻璃管内无水硫酸铜表面的现象是;图中4个洗气瓶装有的试剂请从下列药品中选择:澄清石灰水;酸性高锰酸钾溶液;品红溶液;(可重复选用)试指出各洗气瓶内试剂名称① , ② , ③ , ④ .
(2)检验所得产物含SO2的实验现象为
(3)检验所得产物含CO2的实验现象为 .
(4)写出木炭和浓硫酸反应的化学方程式 .
【答案】
(1)分液漏斗;变为蓝色;品红溶液;高锰酸钾溶液;品红溶液;澄清石灰水
(2)①中品红溶液褪色
(3)③中品红溶液不褪色;④中澄清石灰水变浑浊
(4)C+2H2SO4浓 ![]() 2SO2↑+CO2↑+2H2O
2SO2↑+CO2↑+2H2O
【解析】解:(1)装置中盛浓硫酸的仪器名称是分液漏斗,浓硫酸和碳在加热条件下反应生成二氧化碳、二氧化硫和水,大玻璃管内无水硫酸铜遇到水表面的现象是变为蓝色,验证生成产物成二氧化碳、二氧化硫和水,需要先利用无水硫酸铜检验水蒸气的生成,依次用①检验二氧化硫气体用品红试液褪色证明,②装置利用高锰酸钾溶液除去二氧化硫,③装置利用品红试液检验二氧化硫是否除净,④装置中利用澄清石灰水变浑浊证明二氧化碳的存在,所以答案是:分液漏斗;变为蓝色;品红溶液;酸性高锰酸钾溶液;品红溶液;澄清石灰水.(2)检验二氧化硫气体用品红试液褪色证明,所以答案是:①中品红溶液褪色;(3)③中品红溶液不褪色;④中澄清石灰水变浑浊,证明所得产物含CO2 , 所以答案是:③中品红溶液不褪色;④中澄清石灰水变浑浊;(4)木炭和浓硫酸反应生成二氧化碳、二氧化硫和水,反应的化学方程式为:C+2H2SO4浓 ![]() 2SO2↑+CO2↑+2H2O.所以答案是:C+2H2SO4浓
2SO2↑+CO2↑+2H2O.所以答案是:C+2H2SO4浓 ![]() 2SO2↑+CO2↑+2H2O.
2SO2↑+CO2↑+2H2O.

 永乾教育寒假作业快乐假期延边人民出版社系列答案
永乾教育寒假作业快乐假期延边人民出版社系列答案