��Ŀ����
Fe��Fe2O3����ﹲ27.2g������200mL ϡ H2SO4���ռ�����״����2.24LH2�� ������Һ�еμ� KSCN ��Һ������ɫ��Ϊ�к�����H2SO4����ʹ��Һ�еĽ���������ȫ��ת��Ϊ��������������3 mol/L ��NaOH ��Һ400 mL�����H2SO4 �����ʵ���Ũ��Ϊ�� ��
A�� 0.5mol/L B�� 2mol/L C�� 2.25mol/L D�� 3mol/L
 ����ʦ��Сһ����ʦ������ҵϵ�д�
����ʦ��Сһ����ʦ������ҵϵ�д� ���100�ֵ�Ԫ�Ż�������ϵ�д�
���100�ֵ�Ԫ�Ż�������ϵ�д��±������������г��������ʣ������г������ǵ�һ����Ҫ�ɷ֣������ɷ�δ�г���
��� | �� | �� | �� | �� | �� | �� | �� |
���� | ��Ȼ�� | �� | �� | �մ� | С�մ� | ʯ�� | ͭ�� |
�ɷ� | CH4 | C2H5OH | CH3COOH | Na2 CO3 | NaHCO3 | Ca(OH)2 | Cu |
��1������Ա��Т�~�����Ҫ�ɷֽ��з��ࣨ���ţ�
��һ���������ܵ������_________�����ڵ���ʵ���_________�����ڷǵ���ʵ���__________��
��2��д���۳�ˮ������Ҫ�ɷ�ΪCaCO3)�����ӷ���ʽ__________________________
д���â�����θ���������ӷ���ʽΪ_________________________
�ó���Ģ���Һ����CO2�����ӷ���ʽΪ__________________________
������ˮ����ˮ������Ӧ������ˮ�������Է�Ӧ��
������ʾ���ڲ�ͬ�¶��£�������ˮ������Ӧ�IJ��ﲻͬ���¶ȵ���570 ��ʱ������FeO������570��ʱ������Fe3O4��
ij�о���ѧϰС������ͼ��ʾʵ��װ�ã���ɻ�ԭ������ˮ������Ӧ��ʵ�飬�������о��������ɡ�
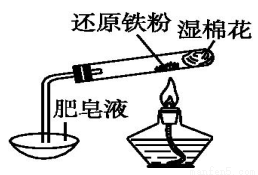
��1������Һ��������____________��
��2����ͬѧΪ̽��ʵ����Թ��ڵĹ��庬����Щ���ʣ�������������ʵ�飺
ʵ���� | ʵ����� | ʵ������ |
�� | ȡ������ɫ��ĩX�����Թ��У��������ᣬ�� | ��ɫ��ĩ���ܽ⣬��Һ����ɫ�����������ݲ��� |
�� | ���Թ��еμӼ���KSCN��Һ���� | ��Һû�г��ֺ�ɫ |
��������ʵ�飬��ȷ��������һ�����ڵ�������______________���ɢڵIJ����������ܷ�ȷ����Ӧ�����в�����Fe3O4��________������ܡ����ܡ���


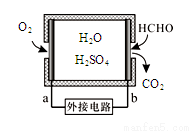

 ˵���д������( )
˵���д������( ) pH��ȵ�CH3COONa��NaOH��Na2CO3������Һ��c(NaOH)��c(Na2CO3)��c(CH3COONa)
pH��ȵ�CH3COONa��NaOH��Na2CO3������Һ��c(NaOH)��c(Na2CO3)��c(CH3COONa)